Iodination [I2/catalyst]
Iodination [I2/catalist] Definition:
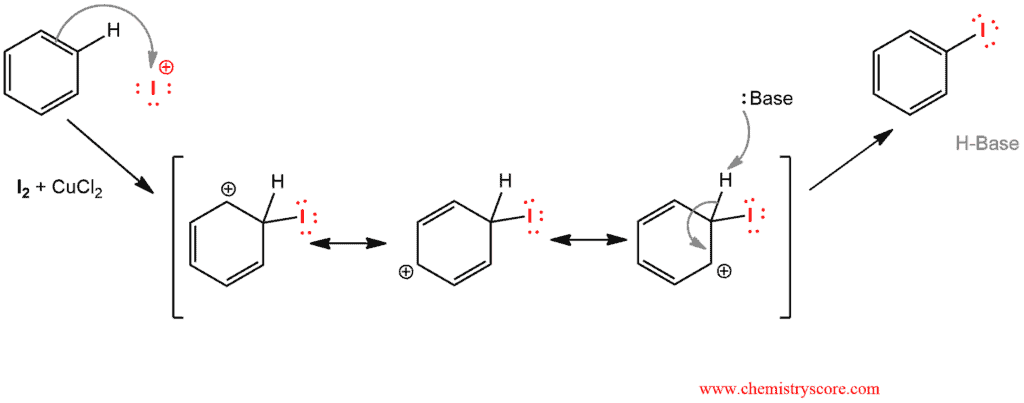
Treatment of an aromatic such as benzene with iodine (I2) and copper (II) leads to the formation of the iodinated benzene by electrophilic aromatic substitution.
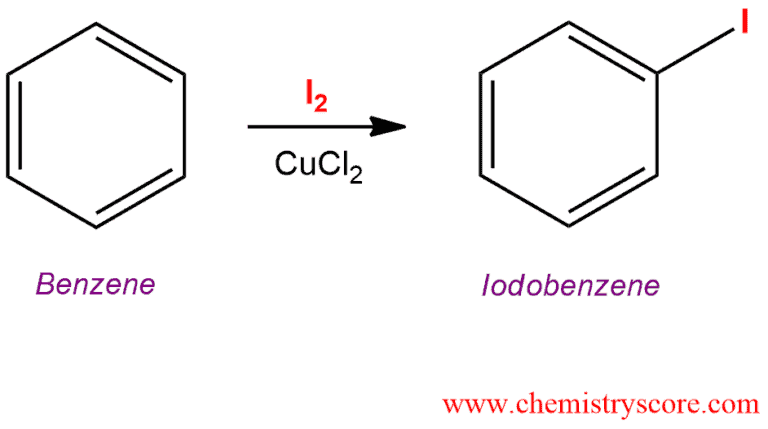
Iodination [I2/catalyst] Explained:
Since iodine is unreactive toward aromatic rings, oxidizing agents such as CuCl2 are used as a catalyst. As well as in any electrophilic aromatic substitution the catalyst is activated first:

And then benzene makes a nucleophilic attack to electrophilic iodine to form sigma complex, or arenium ion, which is resonance stabilized. This cationic intermediate is temporary charged-delocalized which is but with subsequent fast deprotonation regenerates the (now substituted) aromatic ring.