Reduction to amines [LiAlH4]
Reduction to amines [LiAlH4] Definition:
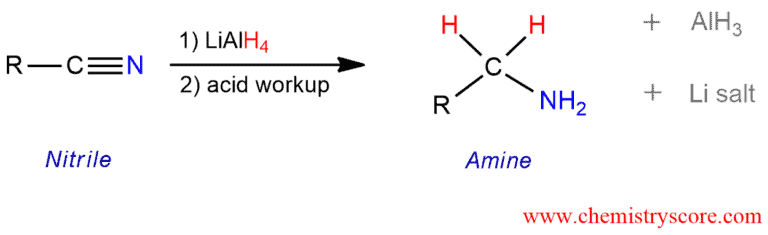
Lithium aluminum hydride, LiAlH4 [but NOT sodium borohydride, NaBH4] can reduce nitriles to primary amines.
Reduction to amines [LiAlH4] Explained:
Treatment of nitriles with a strong hydride reducing agents such as lithium aluminum hydride results in double hydride addition, giving primary amine on aqueous workup. The mechanism of the reaction involves the nucleophilic addition of hydride to the polarized carbon-nitrogen double bond. This reduction requires the addition of two equivalents of hydride from lithium aluminum hydride.
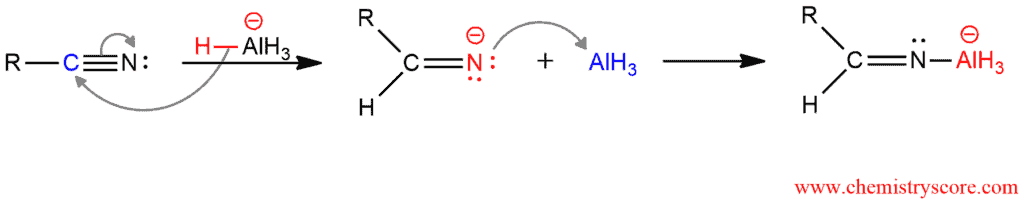
In the first part of the mechanism, the first hydride is added to the electrophilic carbon of the nitrile. This form an intermediate that complexes with the AlH3 which is also formed during the reaction.
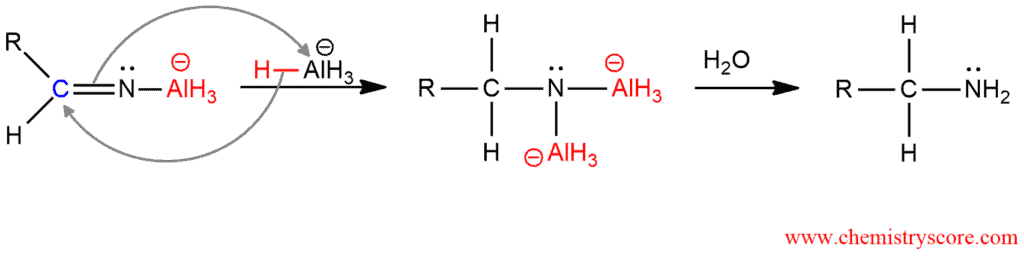
And in the second part of the mechanism, the second equivalent of hydride and the AlH3 is added. The hydrolysis of this dianion forms the primary amine.