Transesterification [basic conditions]
Transesterification [basic conditions] Definition:
Addition of alkoxide to an ester results in a new ester.
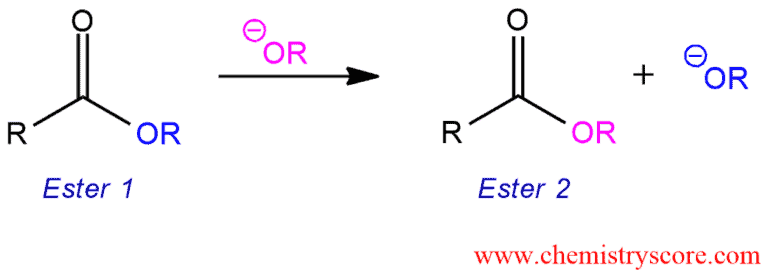
Transesterification [basic conditions] Explained:
Esters react with alcohols in a transformation called transesterification. This allows for the direct conversion of one ester into another without proceeding through the free acid.
Transesterification is a reversible reaction since both the reactants and the products are an ester and an alcohol. To shift the equilibrium, a large excess of the alcohol is usually employed, sometimes in the form of solvent.
This transformation can be achieved with either acid catalysis or base catalysis. The mechanisms of these reactions are analogous to the mechanisms of the corresponding acid- and base-catalyzed hydrolyses of esters to carboxylic acids.
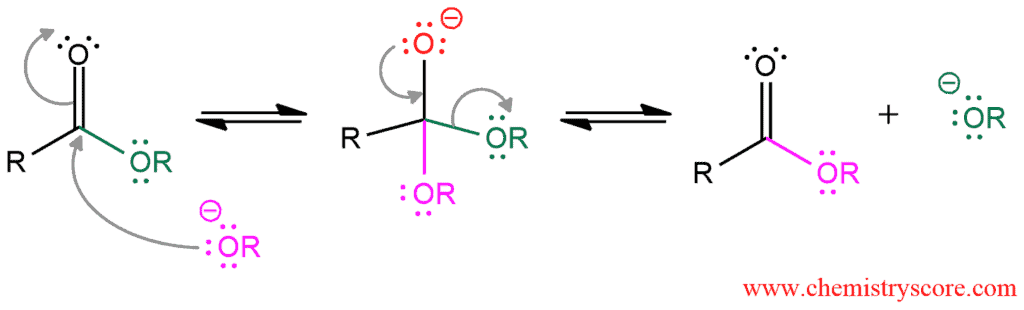
Mechanism of base-catalyzed transesterification
This is a nucleophilic substitution reaction that proceeds by the addition-elimination mechanism. The first step of the mechanism is an addition of a nucleophile, in this case, alkoxide. It attacks the carbonyl carbon and forms a tetrahedral intermediate. Although alkoxide is a poor leaving group it will be readily eliminated in the next step, because the tetrahedral intermediate is sufficiently high in energy that it can expel even a bad leaving group.