Thioether formation RS-
Thioether formation (RS‾) Definition:
Alkyl halides (or tosylates) will react with thiolate anions to form sulfides (or thioether) in an SN2 reaction.

Thioether formation (RS‾) Explained:
Nucleophilic substitution at an sp³ hybridized carbon involves two σ bonds:
- the bond of the leaving group, which is broken
- the bond of the nucleophile, which is formed
This process takes place simultaneously in one-step transformation.
In the transition state, both the leaving group and the nucleophile are partially bonded to the carbon which is sp² hybridized and the molecule is planar. As the reaction proceeds to products, the inversion motion is completed and the carbon returns to the tetrahedral sp³ configuration.
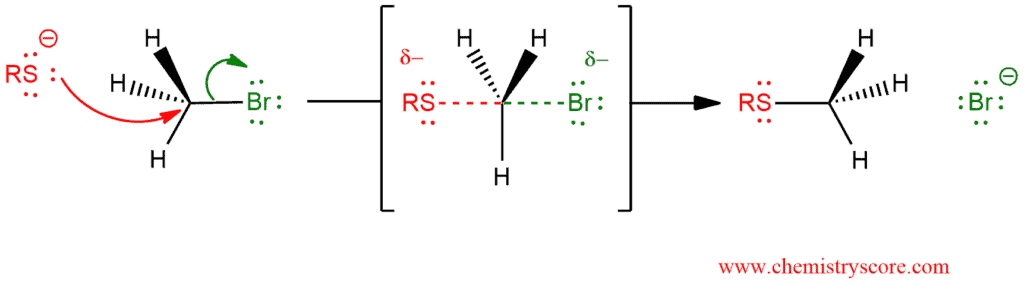
This reaction is the best performed in polar aprotic solvents with primary, possibly secondary alkyl halides.