Suzuki Reaction
Suzuki reaction Definition:
The reaction of an organic halide with an organoboron reagent in the presence of a palladium catalyst.
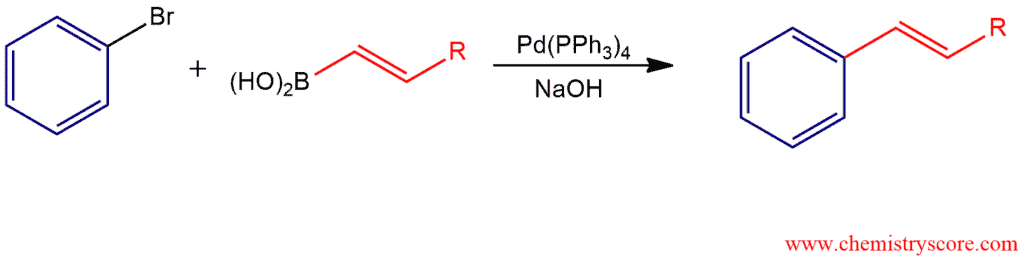
Suzuki reaction Explained:
The Suzuki reaction is a palladium-catalyzed coupling of an organic halide with an organoboron to form a product with a new carbon-carbon bond. As an organohalide is used aryl or alkenyl (vinyl) halide where halide (Cl, Br, I) bonded directly to an sp² hybridized carbon. As a catalyst is used the typically tetrakis(triphenylphosphine)palladium, Pd(PPh3)4, but only the crucial reacting groups around a metal are usually drawn and the other ligands are not specified. And the reaction is carried out in the presence of a base such as NaOH or KOH.
Mechanism of Suzuki Reaction
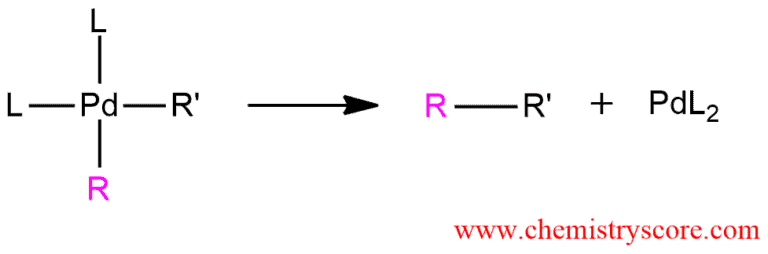
Reaction mechanism with palladium compounds is often multistep. Two of these steps are also common processes which dominate in many reactions of palladium compounds. These processes are an oxidative addition and reductive elimination which implies the addition of reagent, increasing the number of groups around the metal and the elimination of these groups that surround the metal.
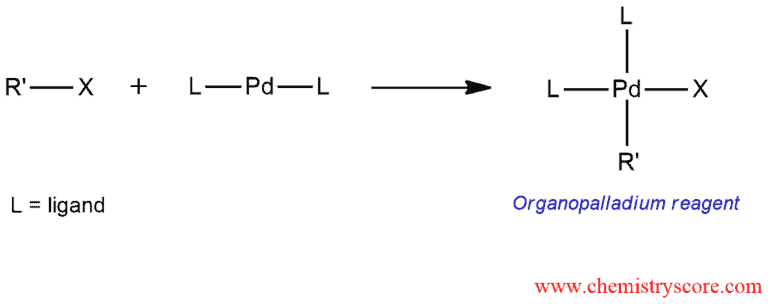
The first step of this mechanism is the formation of an organopalladium compound by oxidative addition. Alkenyl or aryl halide reacts with a palladium catalyst and called oxidative addition because two groups are added to the palladium.
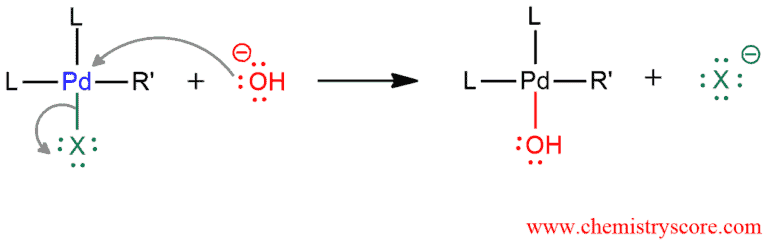
In the second step, a hydroxide which is in solution attacks palladium and the halide is expelled as a good leaving group. This halide is gone and it will not reappear in the rest of this mechanism.
The third step is a nucleophilic attack on an organoboron compound. The boron has three bonds around it and it is sp² hybridized with an empty p orbital. One pair of electrons from the oxygen of OH group will attack the boron and fill the empty p orbital.
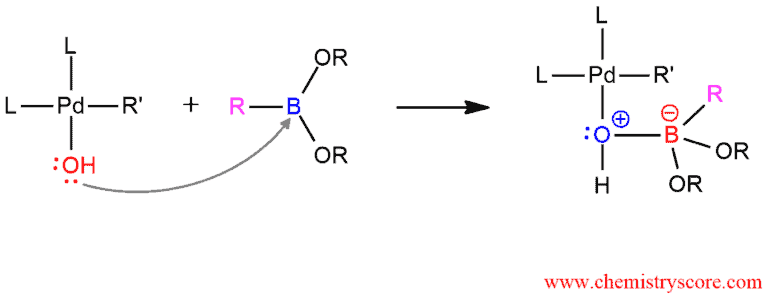
The product which is obtained in the previous step is not stable and because carbon (R) will attack the palladium and the palladium-oxygen bond will break. The positive charge of oxygen will regenerate with two electrons from the broken bond.
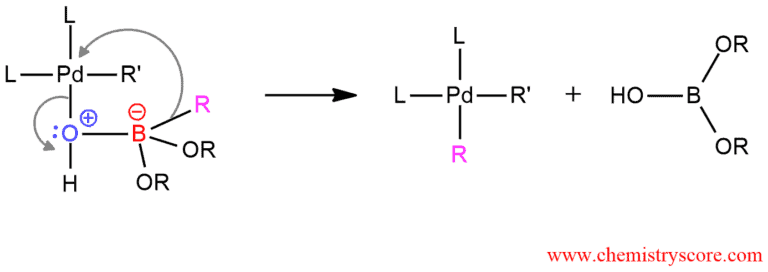
And the last step of this mechanism is the reductive elimination of R’-R where the coupling product is obtained. The palladium catalyst is also regenerated.