Reduction of aldehydes [NaBH4]
Reduction of aldehydes [NaBH4] Definition:
Addition of sodium borohydride, NaBH4 to aldehydes gives primary alcohols (after adding acid).
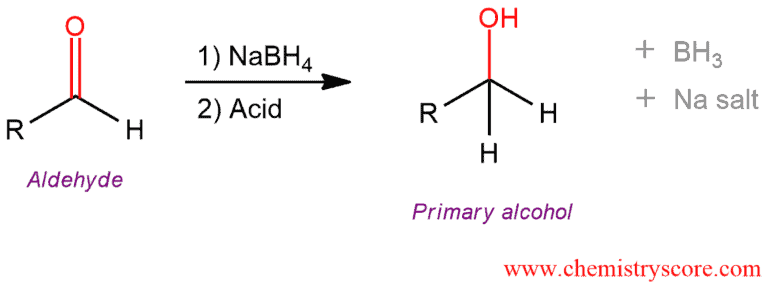
Reduction of aldehydes [NaBH4] Explained:
By reducing the carbonyl group alcohols can be obtained. Primary alcohols are obtained from aldehydes, and secondary from ketones. To reduce the carbonyl group, a hydride reagent is needed such as lithium aluminum hydride, LiAlH4 or sodium borohydride, NaBH4.
Sodium borohydride functions as a source of hydride. Hydride ions are capable of attacking the carbonyl carbon by transferring with their bonding electron pair to generate an alkoxide ion. The alkoxide oxygen is protonated by a solvent. The solvent can be ethanol, methanol, or water. Both of these processes take place simultaneously.
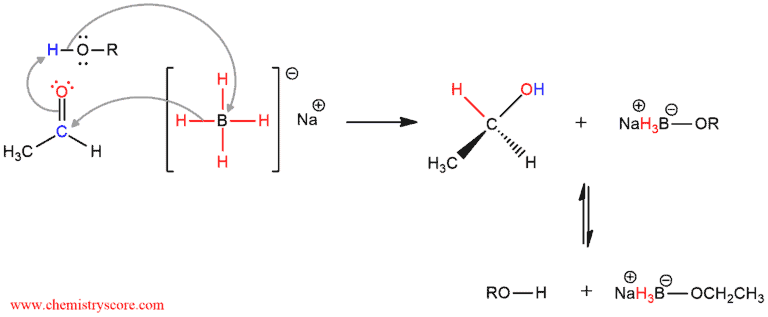
Sodium etoxyborohydride which is obtained can react further with another molecule of aldehyde. Since each H atom of borohydride is involved in the reaction and replaced with one aldehyde molecule, one equivalent of borohydride is consuming four molecules of aldehyde.
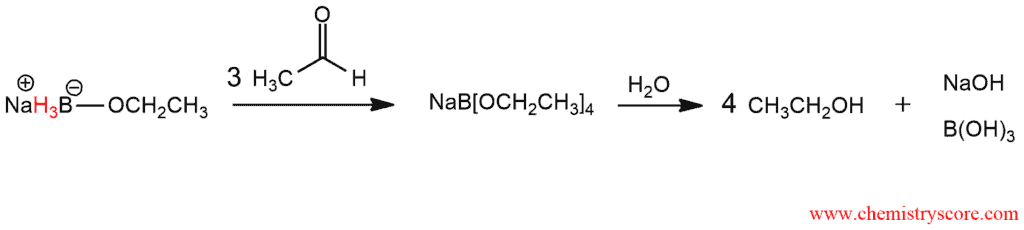
Unlike LAH (lithium aluminum hydride) NaBH4 is less reactive and more selective. Borohydride cannot reduce carboxylic acids, esters, amides and, therefore, selective reduction of aldehydes and ketones in the presence of mentioned other functional groups is possible. Another difference is that in the reduction by NaBH4 hydride transfer to carbon-oxygen double bond is carried out with the previous or simultaneous protonation of carbonyl oxygen by a protic solvent.