Reduction of aldehydes [LiAlH4]
Reduction of aldehydes [LiAlH4] Definition:
Addition of lithium aluminum hydride to aldehydes leads to the formation of primary alcohols (after addition of acid).
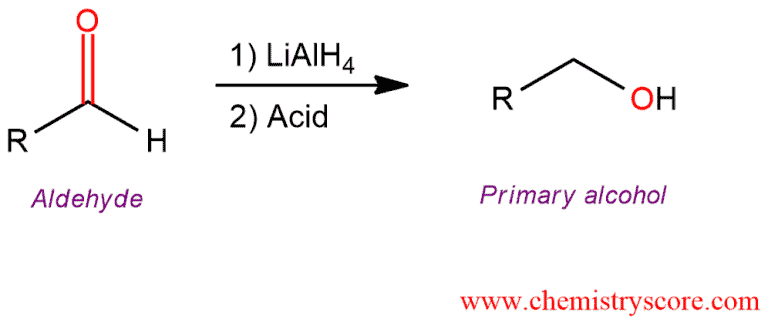
Reduction of aldehydes [LiAlH4] Explained:
Reduction of carbonyl compounds can be carried out by addition of molecular hydrogen or the action of hydride reagents. The carbon-oxygen bond is polarized, the carbon of carbonyl group is electrophilic and the oxygen is nucleophilic. This carbon can react with nucleophilic hydride ion, H‾ from hydride reagents. Two such reagents are sodium borohydride, NaBH4, and lithium aluminum hydride, LiAlH4. They are strong nucleophiles that attack polarized multiple bonds. This attack involves the transfer of hydride ion to a positive atom.
Mechanism of Reduction of Aldehydes with LiAlH4
By transferring one hydride is obtained:
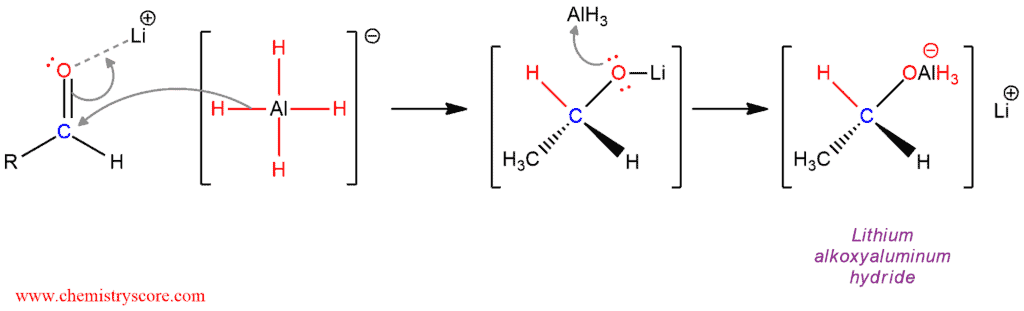
The reduction is carried out in four phases so that all four hydrogen atoms of hydride reagent are involved in the reaction. Each subsequent phase in which the transfer of hydride ion is carried out is slower than the previous one due to the increased number of electronegative -OR alkoxy substituents.
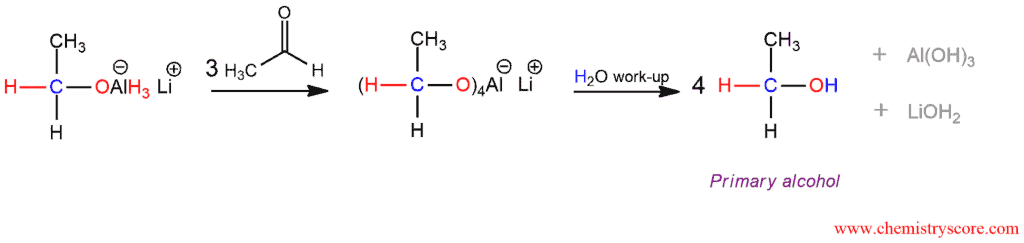
Lithium aluminum hydride abbreviated LAH is a much stronger than sodium borohydride, NaBH4. Because it reacts violently with water a protic solvent cannot be present together with LAH in the reaction flask. It is added in a separate step after treatment with LAH. As a protic solvent can be used water.