Reactions of Diazonium Salts
Reactions of Diazonium Salts Definition:
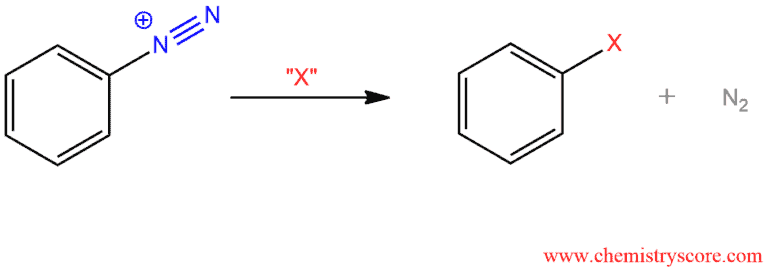
Treatment of diazonium salts with various reagents leads to the formation of a new bond to the aromatic ring and loss of nitrogen (N2).
Reactions of Diazonium Salts Explained:
Aryldiazonium salts are extremely useful because the diazonium group can readily be converted into many other groups which otherwise cannot be easily installed on the aromatic ring. Therefore, the formation of diazonium salts is very important.
Aryldiazonium salts undergo two general reactions:
- Substitution – nitrogen by other group or atom;
- Coupling – diazonium salt with another derivate to form an azo compound.
Substitution reactions
Aryldiazonium salts can react with many different reagents which will replace the diazo group, allowing for a simple procedure for installing a wide variety of groups on an aromatic ring. The nitrogen (N2) is a very good leaving group because it is a gas and once when it formed it leaves the reaction mixture. This makes the reaction irreversible.
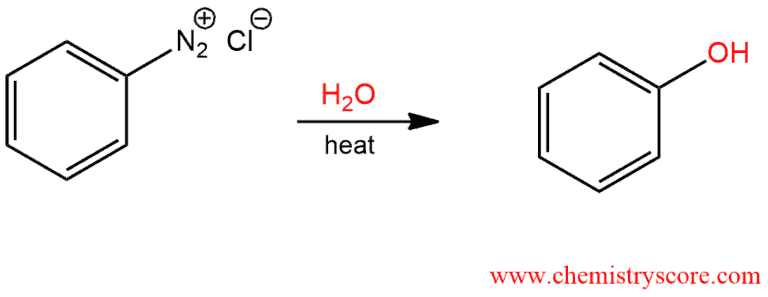
1. Synthesis of phenol – When an aryl diazonium salt is heated in the presence of water, the diazo group is replaced with a hydroxyl group and phenol is formed.
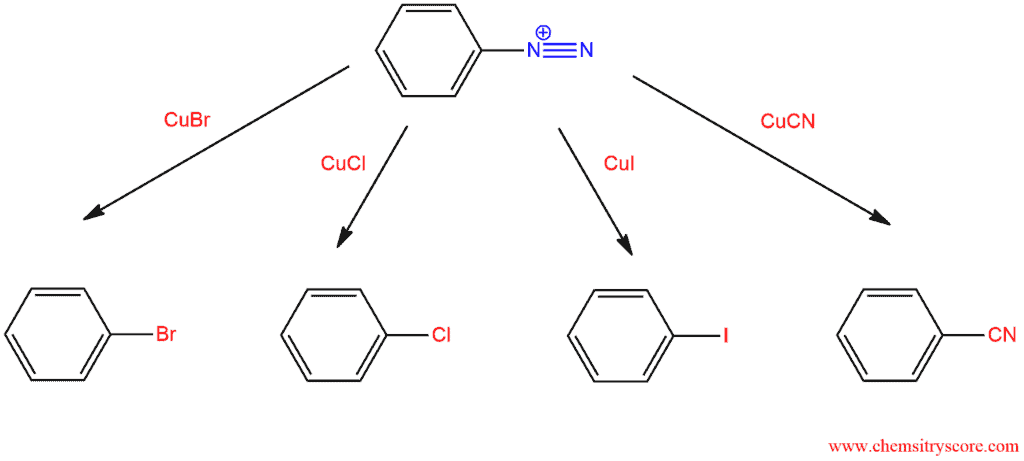
2. Sandmeyer reaction – substitution in which the diazo group is replaced with a halide or a cyano group by using copper salts (CuX).
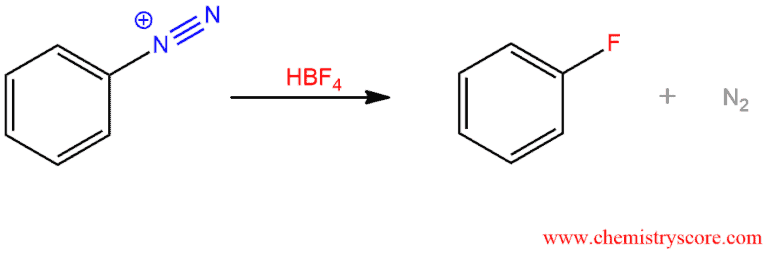
3. Schiemann reaction – a diazonium salt reacts with fluoroboric acid (HBF4) to form aryl fluoride. This is a useful reaction because acyl fluoride cannot be produced by direct fluorination because F2 reacts too violently.
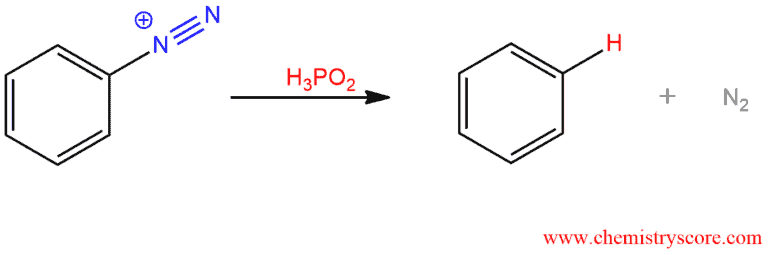
4. Synthesis of benzene – a diazonium salt reacts with hypophosphorous acid (H3PO2) to form benzene.
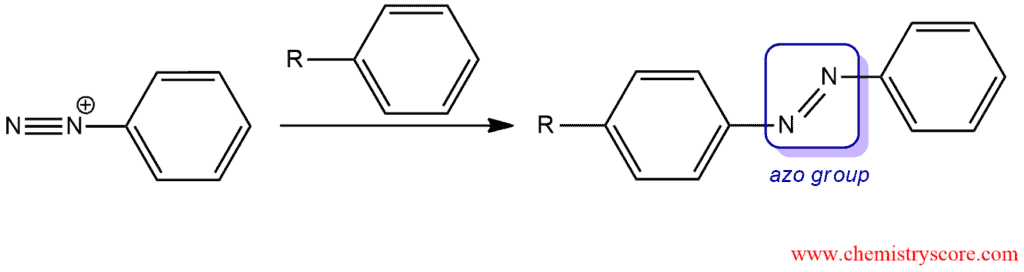
Coupling reactions
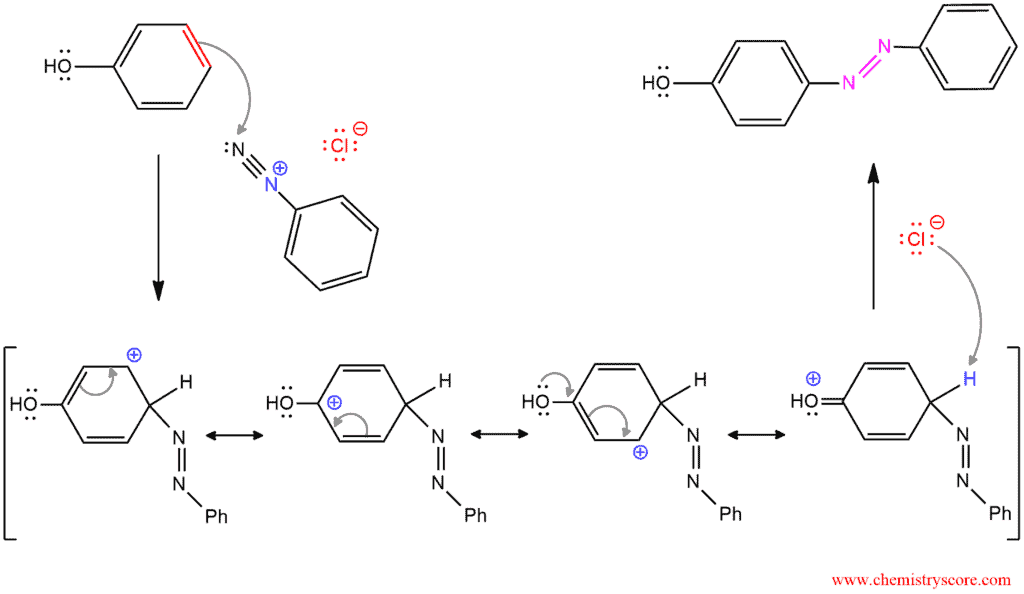
When a diazonium salt is treated with an aromatic compound that contains a strong electron-donor group, the two rings join together to form an azo compound which has a nitrogen-nitrogen double bond.
This process is believed to occur via an electrophilic aromatic substitution reaction, with the diazonium salt acting as the electrophile. The mechanism has two steps. The first step is the addition of the electrophile, the diazonium ion to form a resonance-stabilized carbocation. And the second step is deprotonation of this carbocation.