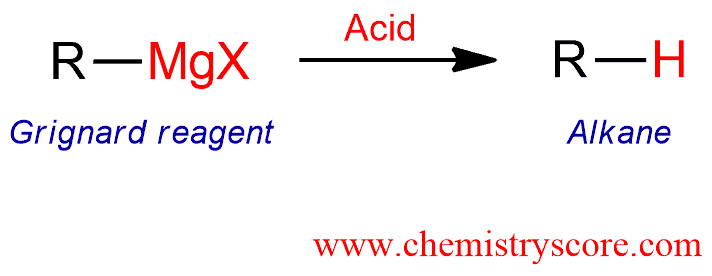
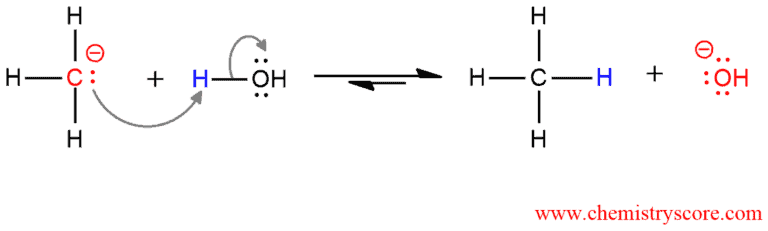Reaction of Grignards with acid [H+]
Reaction of Grignards with acid [H+] Definition:
Treatment of Grignard reagents with acid leads to alkanes. Alternatively, use of deuterated acids leads to the resulting deuterated compounds.
Reaction of Grignards with acid [H+] Explained:
Grignard reagents are rarely isolated. They formed in solution and used immediately in the desired reaction. Because of they sensitive to air and moisture, they must be prepared and handled under rigorously air- and water-free conditions. But when they come in contact with water, they will hydrolize, often violently, to produce a metal hydroxide and alkane. Even any other proton source can protonate Grignard reagent to give alkane. These species are good nucleophiles and strong bases that rapidly abstract a proton from acids, therefore, they are extremely useful in organic synthesis.
The electron pair in the carbon-magnesium bond is used to form a new bond to the proton. Equilibrium favors the products of this acid-base reaction because water is a much stronger acid than the alkane product.