Blog
Protection as silyl ethers
Protection as silyl ethers Definition:
Alcohols can be converted into silyl ethers with trimethylsilyl chloride (TMSCl) or similar silly groups such as t-butyldimethylsilyl chloride (TBDMSCl). This is a useful procedure for the protection of alcohol.
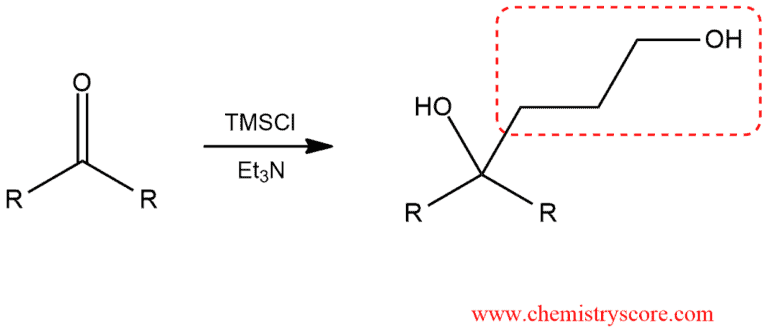
Protection as silyl ethers Explained:
In some cases, we need to protect some group of a molecule to make the desired product. And this is an example of protecting the hydroxyl group of an alcohol with trimethylsilyl chloride via an SN2-like (called SN2-Si) mechanism.
This reaction above would achieve by three following step:
STEP 1: To get a protecting group (trimethylsilyl ether, OTMS) we need to remove the proton from hydroxyl group. This can be achieved with a base, such as triethylamine which will enable that the hydroxyl group functions as a nucleophile to attack the silicon atom and a chloride ion is expelled as a leaving group. The trimethylsilyl ether is formed via the reaction between an alcohol and trimethylsilyl chloride, abbreviated TMSCl.
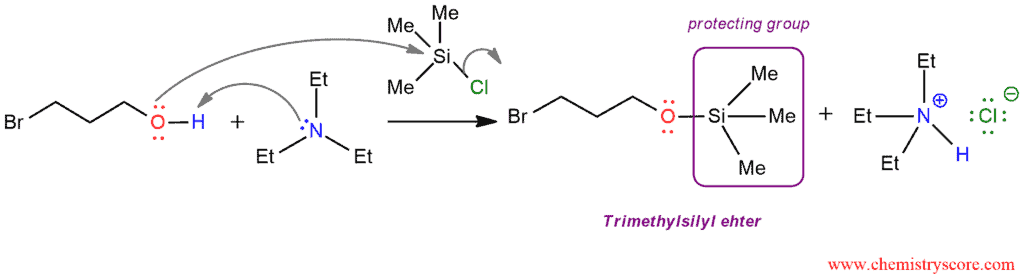
TMSCl is a suitable reagent because if we would use t-butyl chloride a nucleophile could not effectively attack a sterically hindered substrate. The electrophilic center in silicon atom rather than a carbon atom and bonds to silicon atoms are typically much longer than bonds to carbon atoms. This makes the backside of TMSCl a more accessible.
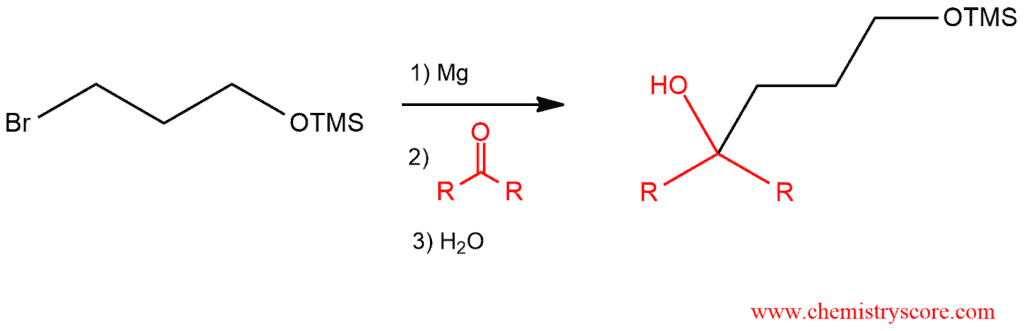
STEP 2: Form the Grignard reagent and perform the desired Grignard reaction which is now enabled because the group of the alcohol is protected.
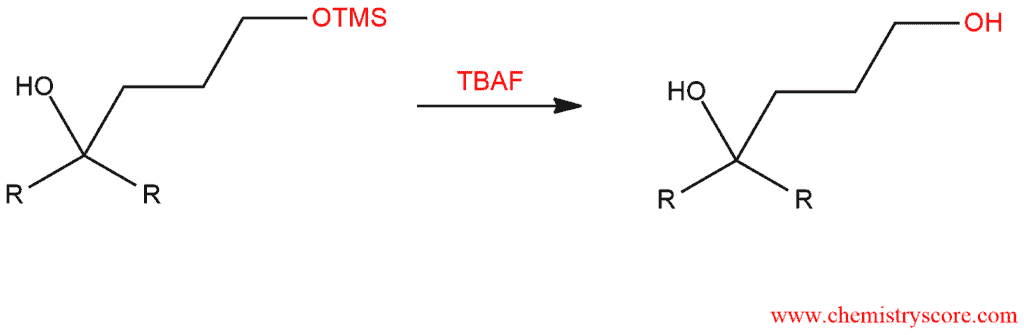
STEP 3: Deprotect by converting the protecting group back into a hydroxyl group. The trimethylsilyl group can be removed with either H3O+ or fluoride ion. A commonly used source of fluoride ion is tetrabutylammonium fluoride (TBAF).