Partial reduction (Lindlar)
Partial Reduction (Lindlar) Definition:
Hydrogenation of the first π bond using the Lindlar catalyst gives cis alkenes.
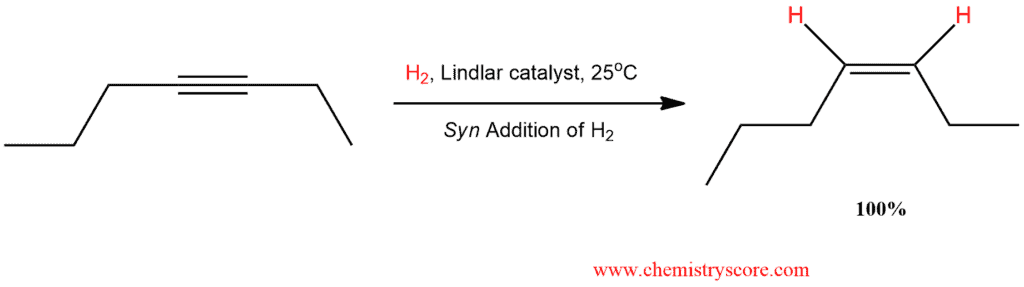
Partial Reduction (Lindlar) Explained:
As we know, using a catalyst such as platinum, palladium or nickel, complete alkynes hydrogenation is achieved. This reaction is practically impossible to stop after the addition of the first hydron molecule in order to obtain alkene. However, with a partially deactivated catalyst, called a poisoned catalyst, it is possible to convert an alkyne into a cis alkene (without further reduction):
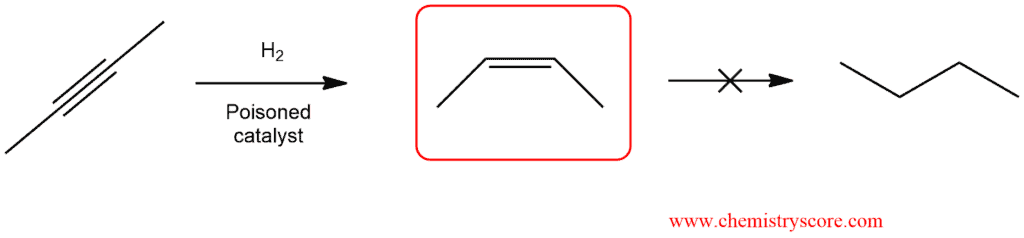
Hydrogenation is a stepwise process that may be stopped at the intermediate alkene stage by the use of a poisoned catalyst, such as the Lindlar catalyst. A poisoned catalyst will catalyze the conversion of an alkyne into a cis alkene, but it will not catalyze the subsequent reduction to form the alkane.
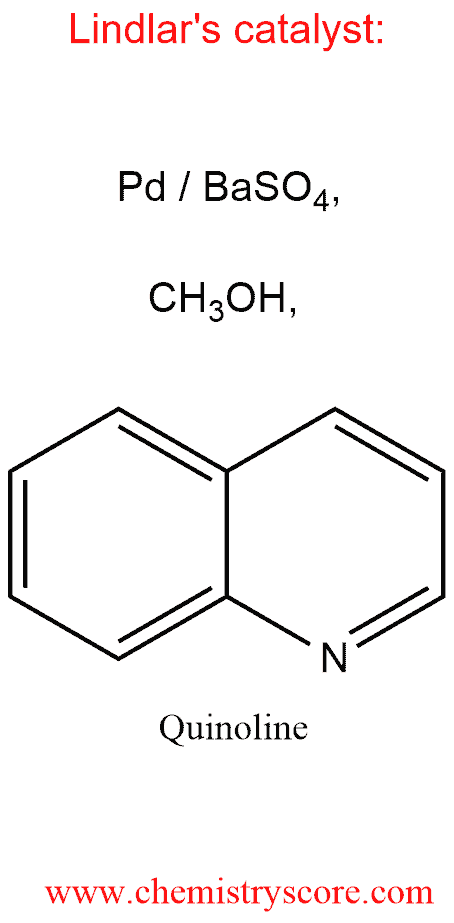
The catalyst is palladium that has been precipitated on barium sulfate and treated with methanol and quinoline. The surface of the metal rearranges a less active configuration than that of palladium on carbon so that only the first π bond of the alkyne is hydrogenated. As with catalytic hydrogenation of alkynes, the addition of H2 is a syn process. As a result, this method affords a stereoselective synthesis of cis alkenes from alkynes.