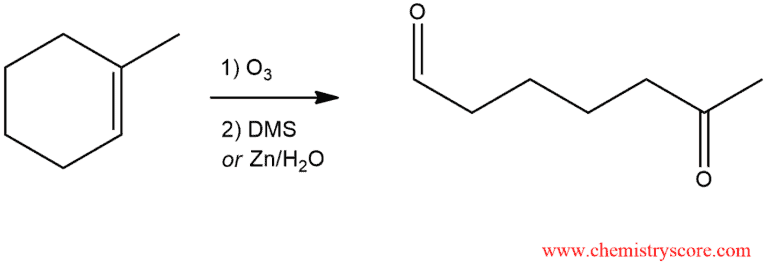
Ozonolysis (Reductive workup)
Ozonolysis Definition:
Ozonolysis is reaction where the double bonds of alkenes are cleaved with ozone (O3). Alkanes form organic compounds in which the multiple carbon-carbon bonds have been replaced by a carbonyl group. The outcome of the reaction depends on the type of multiple bonds being oxidized and the work-up conditions. Reductive work-up conditions are far more commonly used than oxidative conditions.
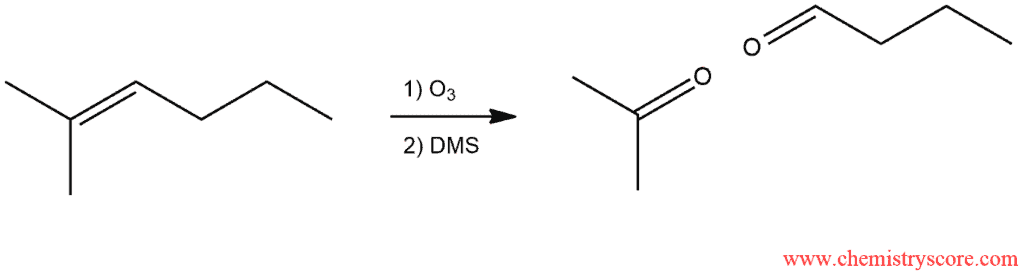
Ozonolysis Explained:
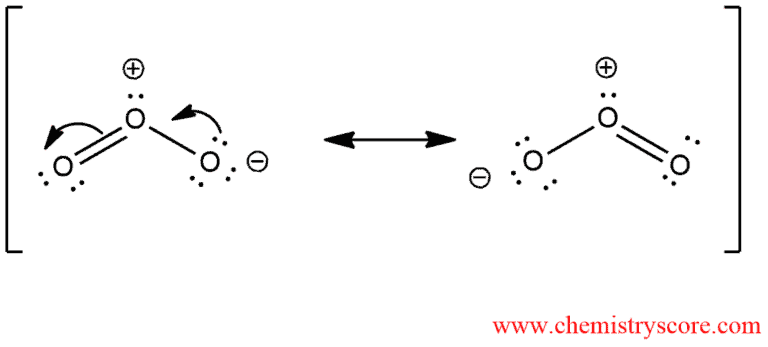
Ozonolysis is the specific reaction in which cleavage occurs and π and σ bond. In this reaction, ozone is mainly used. In order to understand how this reaction occurs, we must first explore the structure of ozone. Ozone is a compound with the following resonance structures:
The gas mixture is passed through a solution of the alkene in methanol or dichloromethane. The first isolable intermediate is a species called an ozonide (or molozonide). The molozonide is unstable and undergoes rearrangement to produce a more stable ozonide:
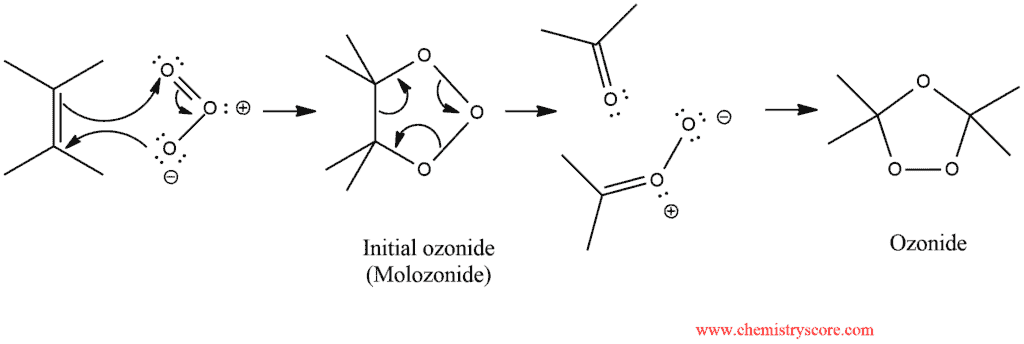
When treated with a mild reducing agent, the ozonide is converted into products:
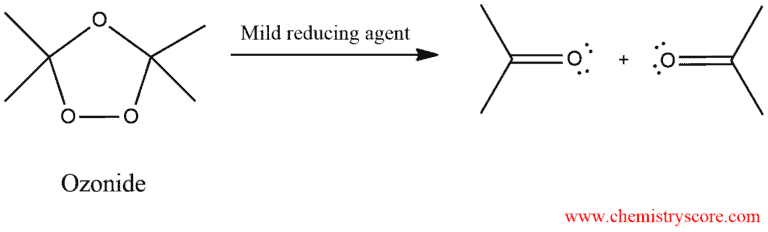
Common examples of reducing agents include dimethyl sulfide (DMS) or Zn/H2O: