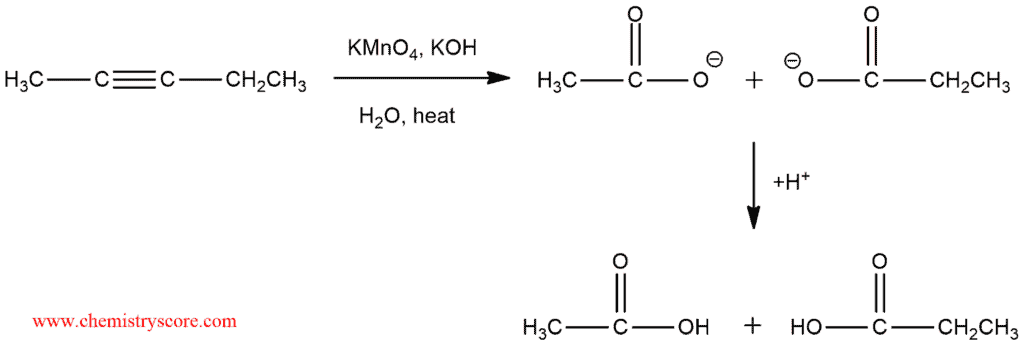
Oxidative cleavage [KMnO4]
Oxidative cleavage Definition:
The oxidative cleavage is the cleavage of the triple bond in the alkyne by an oxidizing agent such as potassium permanganate (KMnO4) which cleave alkynes to give carboxylic acids.
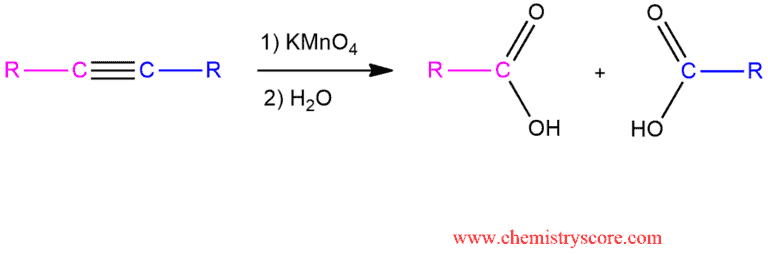
Oxidative cleavage Explained:
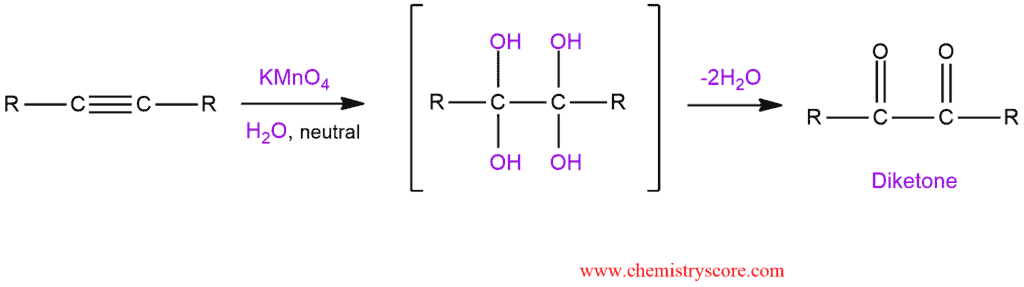
Under neutral conditions. a dilute potassium permanganate solution can oxidize a triple bong into a diketone. The reaction uses aqueous KMnO4 to form a tetrahydroxy intermediate, which loses two water molecules to produce a diketone.
But if potassium permanganate is used under basic conditions or if the solution is heated too much, an oxidative cleavage will take place and two molecules of carboxylic acids will be produced.