Oxidation to carboxylic acids [H2CrO4 + others]
Oxidation to carboxylic acids [H2CrO4 + others] Definition:
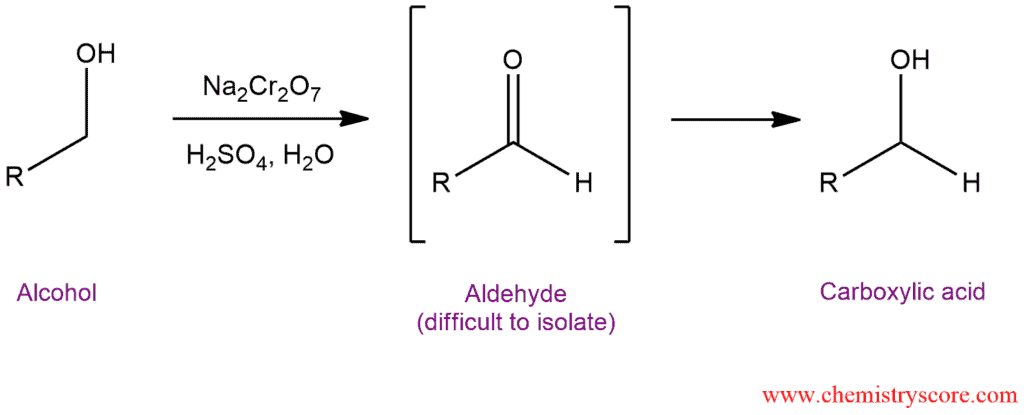
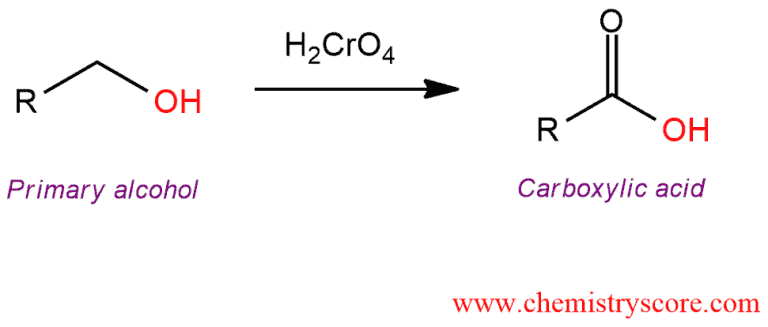
Primary alcohols treated with chromic acid will be converted to carboxylic acid.
Oxidation to carboxylic acids [H2CrO4 + others] Explained:
Oxidation is a reverse process which involves an increase in oxidation state.
The outcome of an oxidation process depends on the starting alcohol. If we have primary alcohol which has two hydrogen atoms at the α position, it can be oxidized twice. The first oxidation yields an aldehyde, and further oxidation of this aldehyde gives the carboxylic acid. Secondary alcohols only have one proton at the α position so they can only be oxidized once, forming a ketone that cannot be oxidized further. Tertiary alcohols do not undergo oxidation because they do not have any protons at the α position.
Chromic acid (H2CrO4) is mainly used to produce a carboxylic acid. It can be formed either from chromium trioxide (CrO3) or from sodium dichromate (Na2Cr2O7) in aqueous acidic solution.
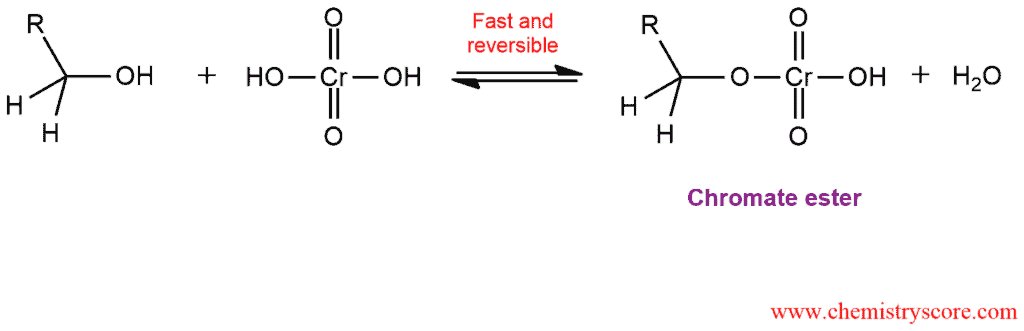
This mechanism consists of two main steps:
STEP 1: Formation of a chromate ester from a primary alcohol and chromic acid.
STEP 2: E2 process form a carbon-oxygen π bond
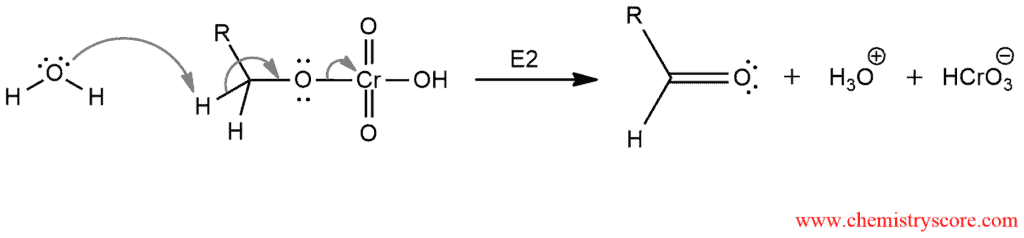
Here obtained an aldehyde which is difficult to isolate. This reaction goes further to carboxylic acid by protonation.