Oxidation to aldehydes [PCC]
Oxidation to aldehydes [PCC] Definition:
Treatment of alcohols with pyridinium chlorochromate (PCC) leads to the formation of the aldehydes.

Oxidation to aldehydes [PCC] Explained:
When a primary alcohol is treated with chromic acid, the oxidation leads to a carboxylic acid and aldehyde which is an intermediate in the reaction is difficult to isolate. In order to produce the aldehyde as the final product, it necessary to use reagent which will react only with alcohol. It would be a more selective oxidizing reagent such as pyridinium chlorochromate (PCC) which is formed from pyridine, chromium trioxide, and hydrochloric acid.
This mechanism consists of following steps:
STEP 1. Attack of oxygen atom on the chromium to form the Cr-O bond.
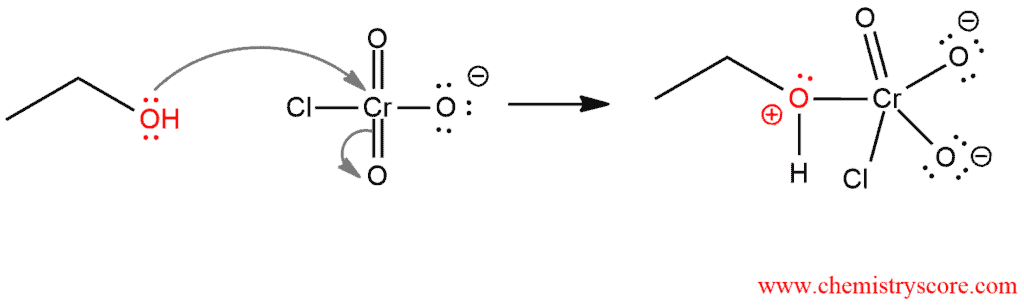
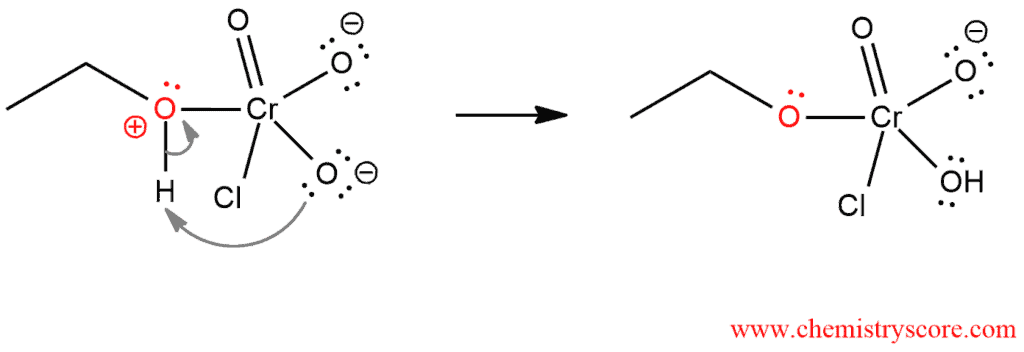
STEP 2. Proton on the positively charged oxygen is transferred to one of the oxygens of the chromium.
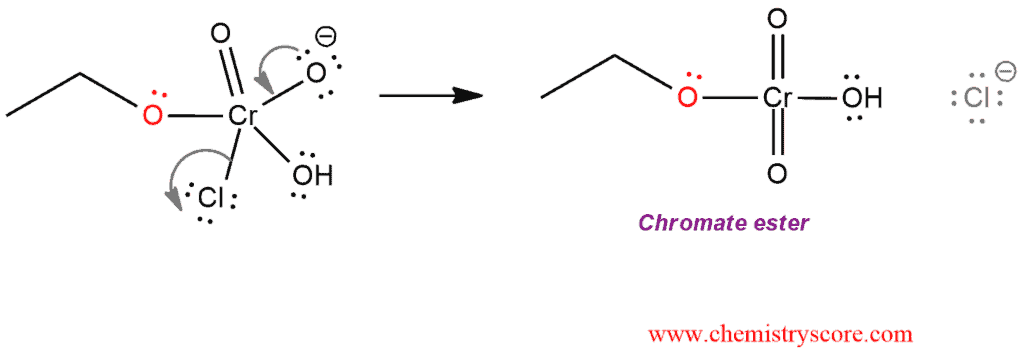
STEP 3. Elimination of chloride leads to the formation of chromate ester.
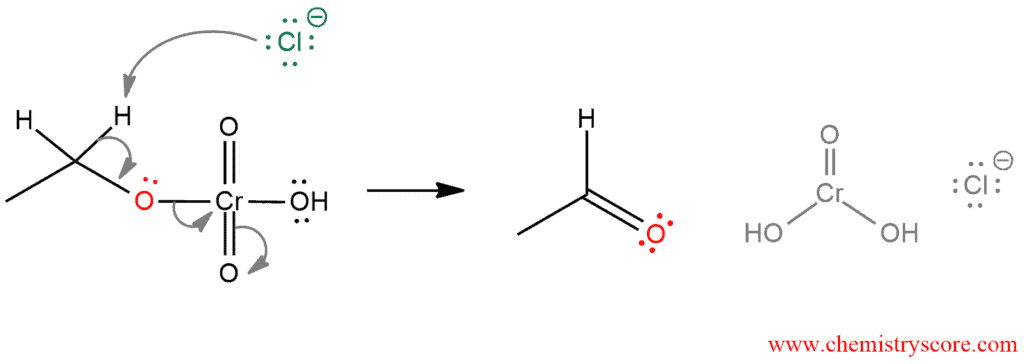
STEP 4. The carbon-oxygen double bond is formed when a base (chloride ion) removes the proton on the carbon in adjacent to the oxygen. The electrons from the C-H bond move to form the C-O bond, and in the process break the O-Cr bond, chromium(VI) is reduced to chromium(IV).