Halogenation [Cl2, Br2, I2]
Halogenation Definition:
In the same way as alkenes, alkynes are also observed to undergo halogenation. The one major difference is that alkynes have two π bonds rather than one and can, therefore, add two equivalents of the halogen to form a tetrahalide (tetrahaloalkanes).
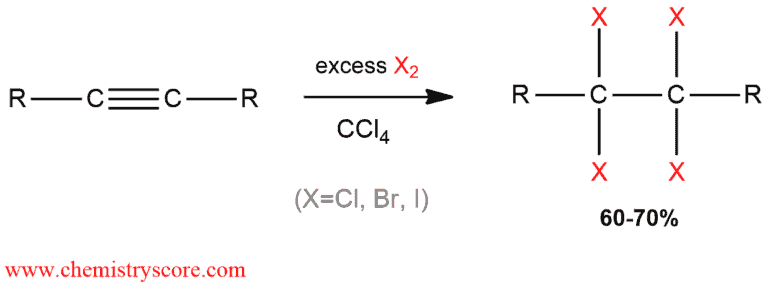
Halogenation Explained:
Halogenation like most addition reaction of alkynes can also take place once or twice. Electrophilic addition of halogens to alkyne proceed through the intermediacy of isolable vicinal dihaloalkenes, the products of a single anti addition.
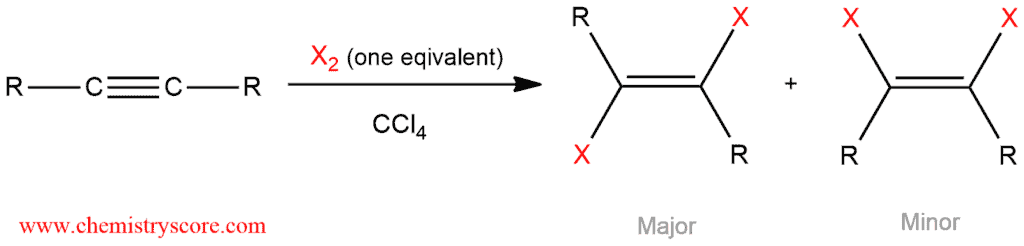
The mechanism of alkyne halogenation is not entirely understood.