Grignard formation [alkenyl halides]
Grignard formation [alkenyl halides] Definition:
Treatment of an alkenyl halide with magnesium metal leads to the formation of the Grignard reagent.
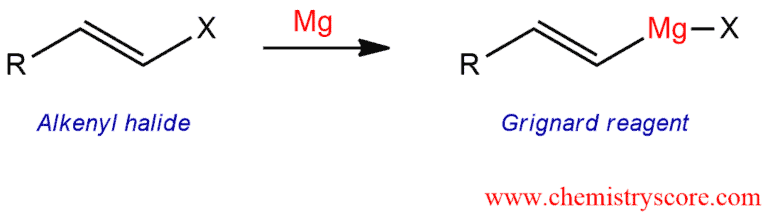
Grignard formation [alkenyl halides] Explained:
A Grignard reagent has a formula RMgX where X is a halide, and R is an alkyl or aryl (based on a benzene ring) or in this case alkenyl group. As these reagents belong to the group of organometallic reagents, they are also called organomagnesium reagents.
A Grignard reagent is formed by the reaction between alkyl halide (haloalkane or halogenalkane) and magnesium in ether (ethoxyethane or diethyl ether) or tetrahydrofuran, THF (oxacyclopentane). For this reaction cannot be used a protic solvent because Grignard reagent is very reactive and in the presence of water or alcohol they will immediately grab hydrogen from water or alcohol and deactivated itself.
In Grignard reagents, a carbon atom of an organic group is bound to a metal. Carbon is more electronegative than magnesium, so the carbon atom withdraws electron density from magnesium via induction. This gives rise to a partial negative charge (δ-) on the carbon atom. In fact, the difference in electronegativity between carbon and magnesium is so large that the bond can be treated as ionic.
Magnesium is in the second column of the periodic table and it has two valence electrons. This is very reactive metal and it will give its two electrons to the carbon atom. The C-Br bond will break because Br is more electronegative than carbon. The alkenyl ion is obtained. So the carbon has a negative charge. Magnesium lost two electrons and it has a plus two charge. And bromine took electrons from C-Br bond and it has a negative charge. The carbon immediately bonds with positively charged magnesium ion because it is not stable.
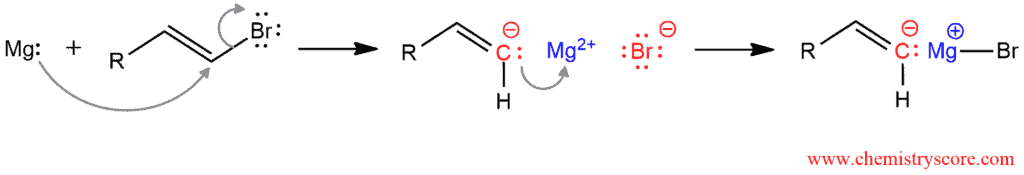
Grignard reagents are strong bases and they react with acidic hydrogens (such as alcohols, water, and carboxylic acids). These species are also good nucleophiles which can react with electrophiles such as carbonyl compounds (aldehydes, ketones, esters, carbon dioxide etc) and epoxides. As such, they are extremely useful in organic chemistry.