Gabriel Synthesis of amines
Gabriel Synthesis of Amines Definition:
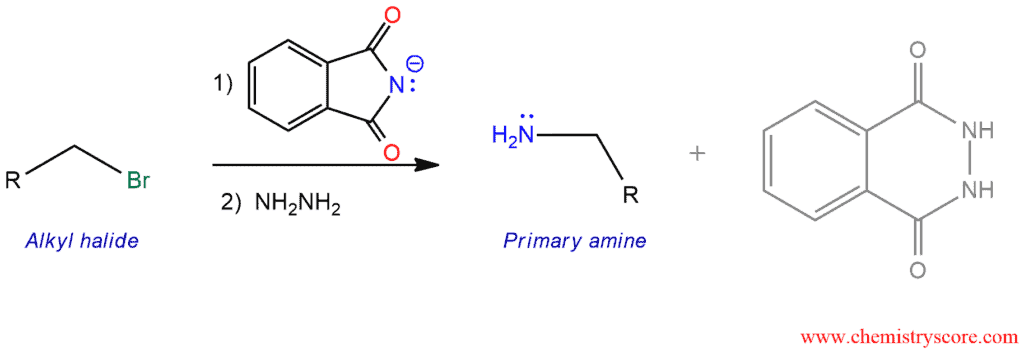
The Gabriel synthesis is a method for making primary amines through an SN2 reaction of a phthalimide with an alkyl halide followed by cleavage with hydrazine.
Gabriel Synthesis of Amines Explained:
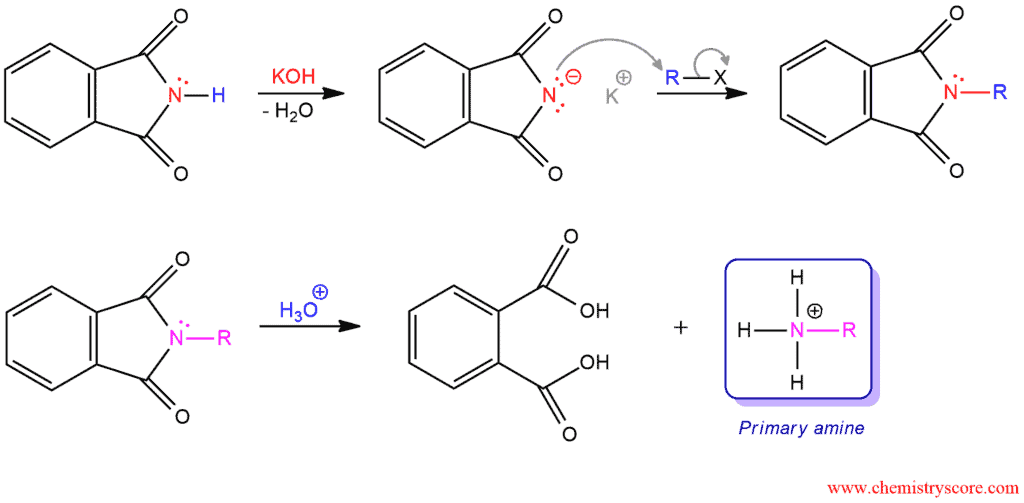
The Gabriel synthesis is a nonreductive method for preparing primary amines with potassium phthalimide which is prepared by treating phthalimide with potassium hydroxide. The acidity of NH group is much greater than that of an ordinary amide because the nitrogen in the imide is adjacent to two carbonyl functions. Therefore, the deprotonation will be easily performed. The resulting anion function as a nucleophile and is readily alkylated by SN2 reaction to form a carbon-nitrogen bond. This reaction works best with primary alkyl halides and sometimes with secondary, but tertiary cannot be used. Then this product can be hydrolyzed by acid or base.
Hydrolysis of Amides Under Acidic Conditions
Acidic conditions are more common than basic conditions. An ammonium ion is produced which must be treated with a base to remove the charge and formed an amine.
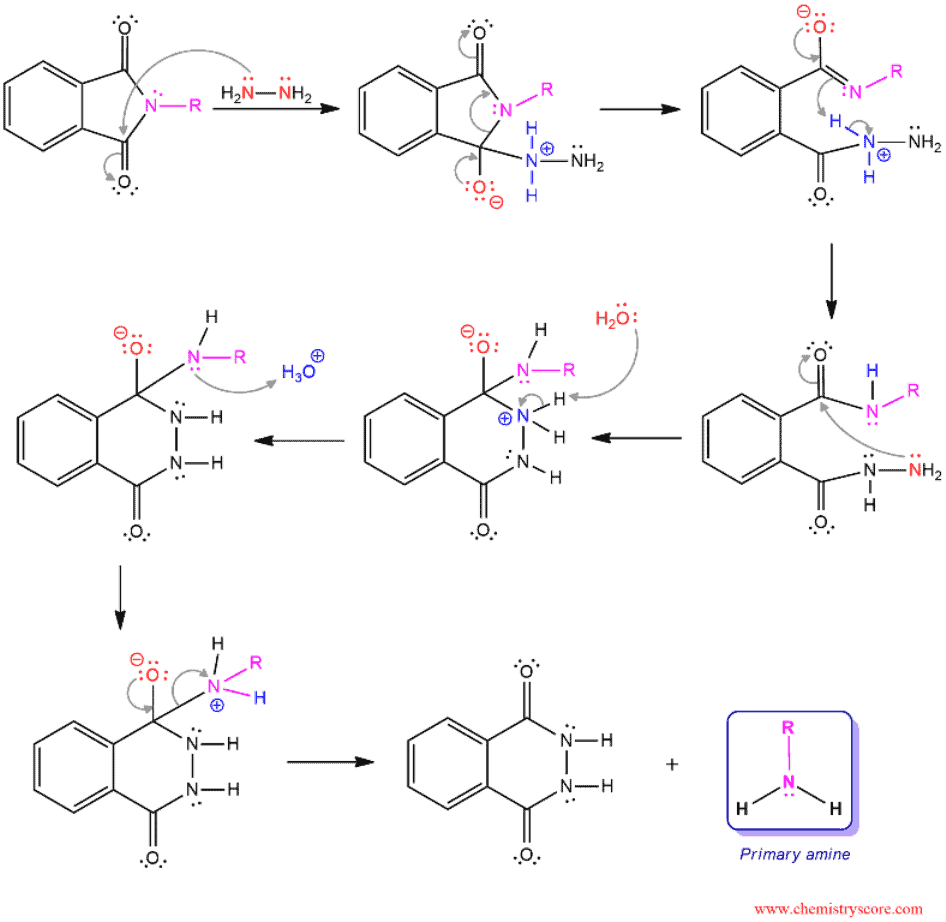
Hydrolysis of Amides Under Basic Conditions
This is one of many predicted mechanisms. Hydrazine function as a nucleophile and attacks both carbonyl functions with both nitrogen atoms. Primary amine and phthalimide hydrazide are obtained as products of this mechanism.