Friedel Crafts alkylation [R-X plus catalyst]
Friedel Crafts alkylation [R-X plus catalyst] Definition:
In Friedel-Crafts alkylation, treatment of benzene with an alkyl halide and a Lewis acid (AlCl3 or FeCl3) forms an alkylbenzene. This reaction is an alkylation because it results in the transfer of an alkyl group from one atom to another (from X to benzene).
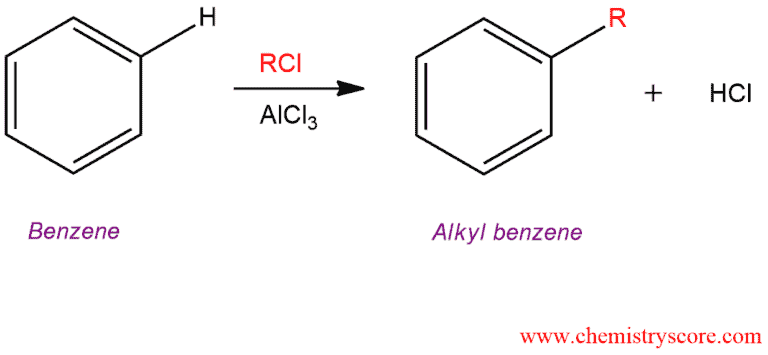
Friedel Crafts alkylation [R-X plus catalyst] Explained:
The Friedel-Crafts alkylation forms new carbon-carbon bonds by the installation of an alkyl group on an aromatic ring.
The reactivity of alkyl halides increases with the polarity of the carbon-halide bond in the order: RI < RBr < RCl < RF
Typical Lewis acids are FeCl3, AlCl3, and AlBr3.
Mechanism of Friedel-Crafts alkylation
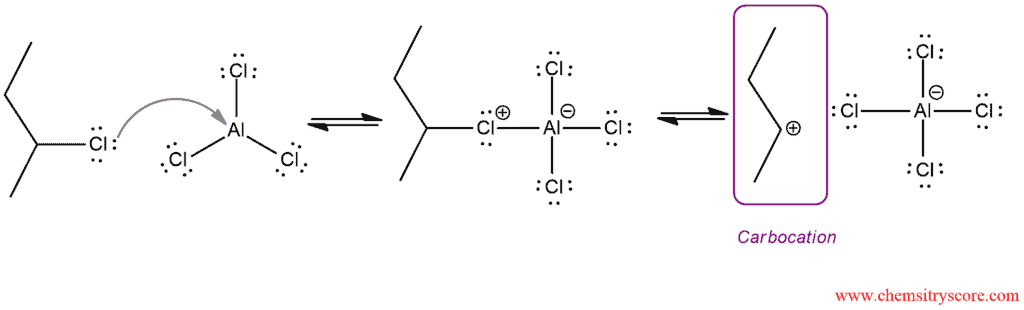
Step 1. Alkyl halide activation and formation of carbocation which is an excellent electrophile
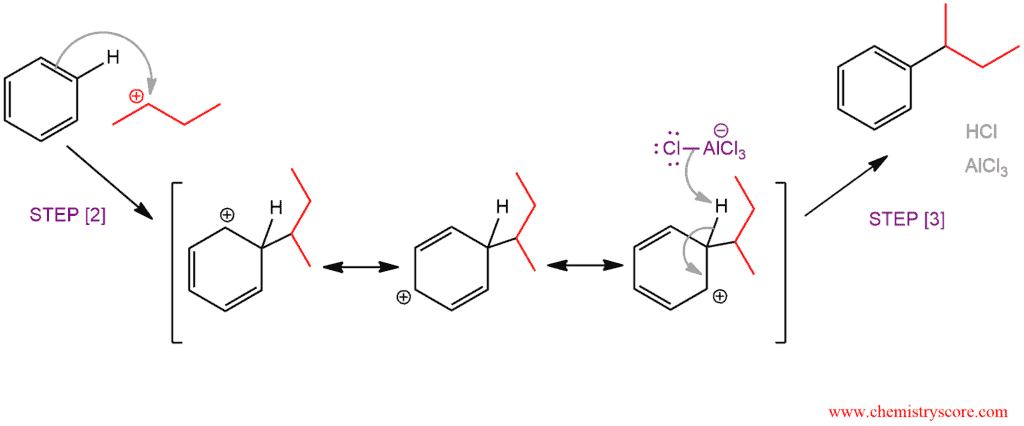
Step 2. Nucleophilic attack of benzene to carbocation and form an intermediate sigma complex
Step 3. Deprotonation of sigma complex to restore aromaticity
This is an example for secondary and tertiary halides but with primary halides, it is slightly different. Primary alkyl halides are not converted into carbocations since primary carbocations are extremely high in energy. In this case, the electrophilic agent is presumed to be a complex between alkyl halide and catalyst.
Friedel-Crafts alkylation can be carried out with any starting material that functions as a precursor to a carbocation, such as alcohol or alkane. And also can be used to fuse a new ring onto the benzene nucleus by an intramolecular Friedel-Crafts alkylation.
Disadvantages of Friedel-Crafts alkylations are overalkylation and skeletal rearrangements by both hydrogen and alkyl shifts which do not case with Friedel-Crafts acylation.