Formation via Grignard and CO2
Formation via Grignard and CO2 Definition:
Grignard reagents will add to carbon dioxide, forming carboxylate salts. After acidic work-up, carboxylic acids are formed.
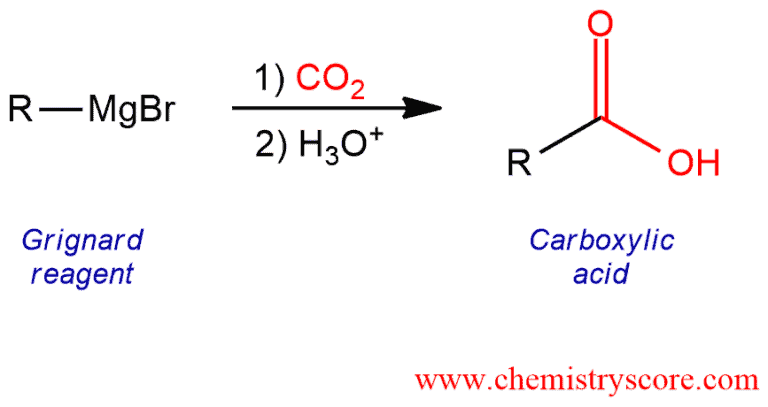
Formation via Grignard and CO2 Explained:
Carboxylic acids can be prepared by treating a Grignard reagent with carbon dioxide (usually in the solid form known as “dry ice”). The product of this carbonation process is a carboxylate salt, which upon protonation by aqueous acid yields the carboxylic acid.
Mechanism of preparation of carboxylic acids via Grignard compound
In the first step, the Grignard reagent attacks the electrophile center of carbon dioxide, generating a carboxylic ion.
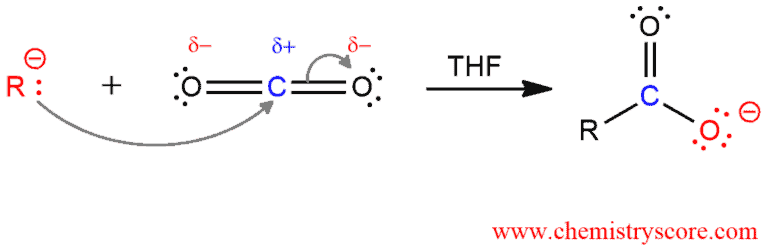
In the second step, protonation of carboxylic ion leads to the carboxylic acid.
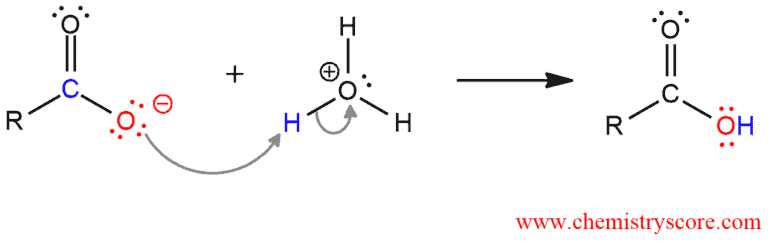
These two steps occur separately because otherwise, the first step would take place before the other.