Formation of organolithium reagents
Formation of the organolithium reagents Definition:
Addition of lithium metal (2 equivalents) to an alkyl or alkenyl halide results in the formation of the organolithium reagents.

Formation of the organolithium reagents Explained:
Organolithium reagents have a formula RLi where R can be alkyl, aryl, allyl, or benzyl group. But they are typically prepared by reaction of an alkyl halide with lithium metal suspended in ether (diethyl ether) or tetrahydrofuran, THF. The reactivity of the alkyl halides increases in the order Cl < Br < I, but F is the relatively unstable and they cannot use in these reactions.
Alkyl halide will react with two equivalents of lithium; one lithium will replace the halide and the other one will bind to the bromine to give lithium halide, LiX.
Lithium is in the first column of the periodic table and it has one valence electron. But two equivalents of lithium have 2 valence electrons which can donate to the carbon atom of methyl bromide. This bond, carbon-bromide will break because bromine is more electronegatively than carbon. The carbanion is obtained that has three hydrogen atoms and alone pair. So the carbon has a negative charge. Both of the lithiums lost their electrons and become positively charged. And bromine took electrons from C-Br bond and it has a negative charge. Bromide ion will bind one lithium ion.
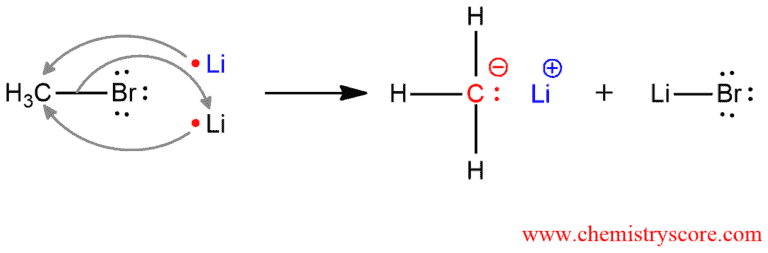
Organolithium reagents contain a carbon atom bonded to a metal. Lithium is more electropositive (less electronegative) than carbon and it donated electron density towards carbon so that carbon bears a partial negative charge. Because of this electropositively, bond carbon-lithium is polar and therefore organolithium reagents are very reactive. In reaction, they behave more or less like carbanions. It is therefore said that they are strong nucleophiles which can react with electrophilic carbon atoms to form new carbon-carbon bonds. These reactions are very valuable in forming the carbon skeletons of complex organic molecules.