Enolate formation
Enolate formation Definition:
When treated with a strong base, the α position of a ketone or aldehyde is deprotonated to give a resonance-stabilized intermediate called an enolate ion or enolate.
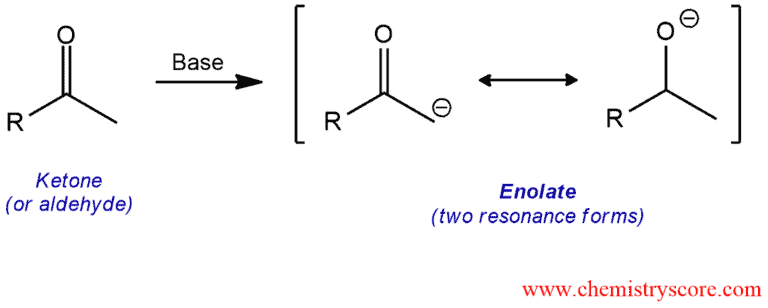
Enolate formation Explained:
The structure of carbonyl group—a double bond which is highly polar—makes these compounds subject to the following reactions:
- Addition of an electrophile on the Lewis basic oxygen, usually by protons
- Attack of nucleophile on the carbon
- Reactions with α-carbon and α-hydrogen (this is the carbon that is next to the carbonyl group and its hydrogens).
In the presence of the strong base, α-hydrogens can be removed. The anions that result are known as enolate ions or simply enolates.
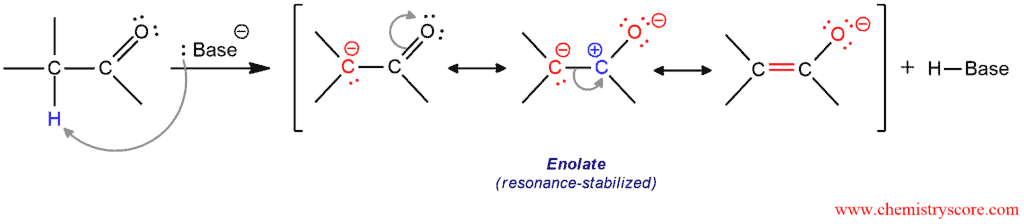
The resonance hybrid possesses partial negative charges on both carbon and oxygen and electrophile can attack at either position. That makes enolates ambident nucleophiles. When the oxygen atom attacks an electrophile, it is called O-attack; and when the α carbon attacks an electrophile, it is called C-attack. The most common reactions that take place are the following:
- Alkylation – SN2 reaction with alkyl halides
- Keto-enol tautomerisation – protonation of oxygen to produce an unsaturated alkenol or simply enol which is unstable and rapidly isomerize back to the original ketones
- Haloform reaction – the reaction of a methyl ketone with halides in the presence of hydroxide ions to give a carboxylate ion and a haloform.