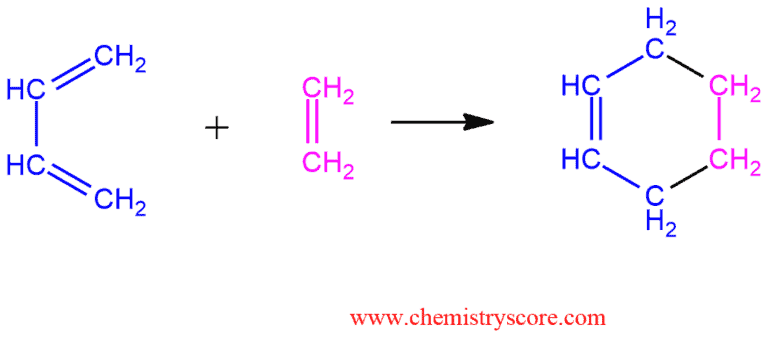
In Diels-Alder reaction (or Diels-Alder cycloaddition), the atoms at the ends of the diene add to the dienophile double or triple bond (alkene or alkyne), thereby closing a ring (product is cyclohexene). The new bonds form simultaneously and stereospecifically. It does not include even an intermediate, it all happens in one step. This reaction is also called [4+2] cycloaddition because reaction takes place between four conjugated atoms containing four π electron react with a double bound containing two π electrons.
The Diels-Alder reaction takes place in one step. Both new carbon-carbon σ bonds and the new π bond form simultaneously, just as the three π bonds in the starting materials break. For such one-step reactions, we say they are concerted.
The mechanism of this reaction should not present a major problem in understanding. You just need to get closer diene to dienophile and add sp² carbon atoms of diene to sp² carbon atoms of dienophile. The arrows can be drawn proceeding in a clockwise fashion or vice versa. The end result is the same.
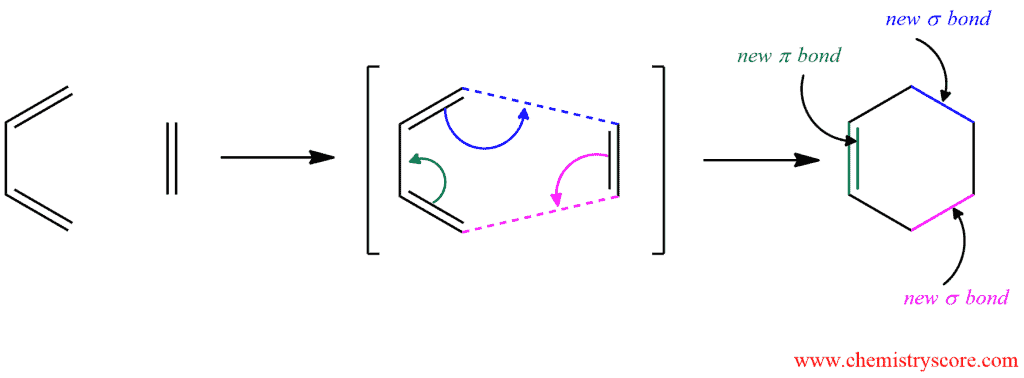
In this example, the simplest representatives of diene and alkene are used, 1,3-butadiene and ethene. But what happens if we these compounds are substituted? Then we must take care of stereochemistry!
Stereochemistry of the dienophile
When the dienophile doesn’t contain any substituents, the reaction is slow and the yield is low. But if dienophile contains an electron-withdrawing substituent such as a carbonyl group, the reaction will proceed more rapidly and with a much higher yield. These substituted dienophiles lead to the formation of chiral center(s). And because we must think about stereochemistry.
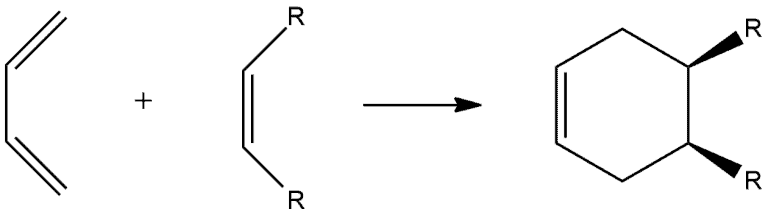
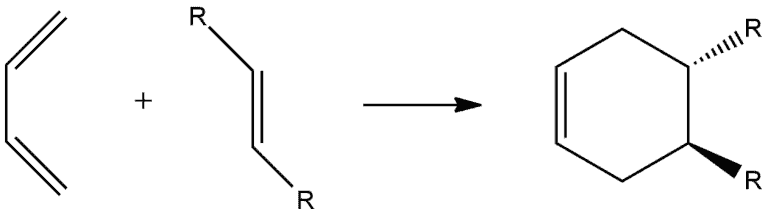
The stereochemistry at the original double bond of the dienophile is retained in the product. Specifically, a cis dienophile produces a cis disubstituted ring, and a trans dienophile produces a trans disubstituted ring.
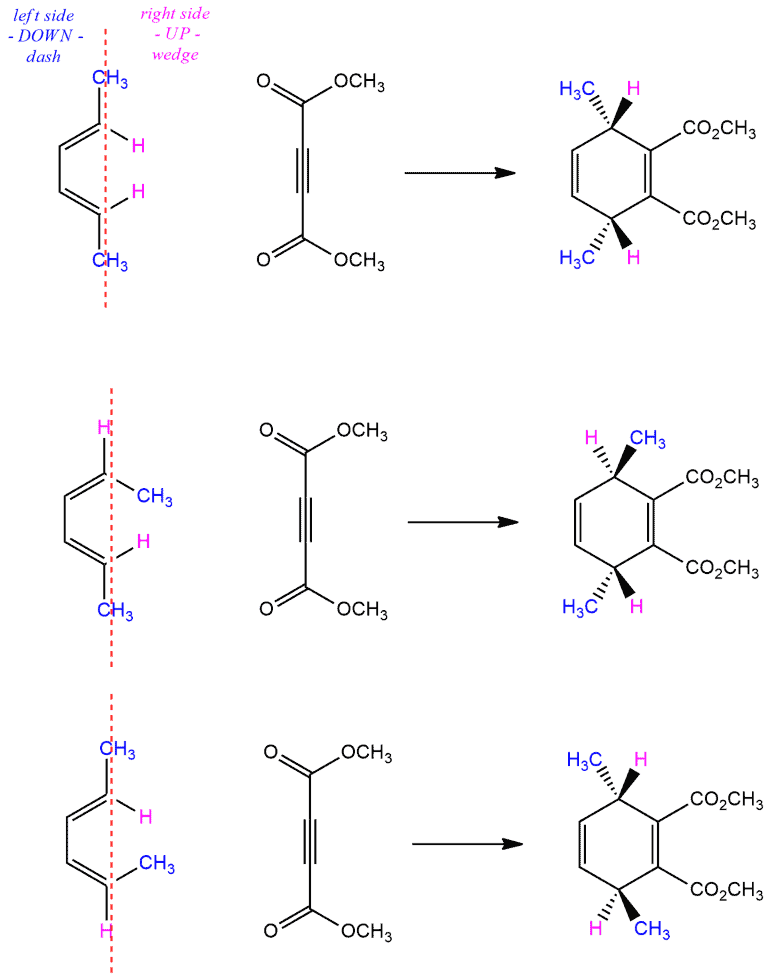
Look at the following example. We have dienophile with two R groups which are cis to each other, so they are on the same side. Carbons of dienophile go from being sp² hybridized to being sp³ hybridized forming chiral centers. Since we have concerted movement of electrons, these two R groups end up on the same side.
If we look our dienophile and think about the groups on the left and the right side of the line, the groups on the right side will be always up in the product (drawn on the wedges). And groups on the left side of the line will be always down in the product (drawn on dashes).
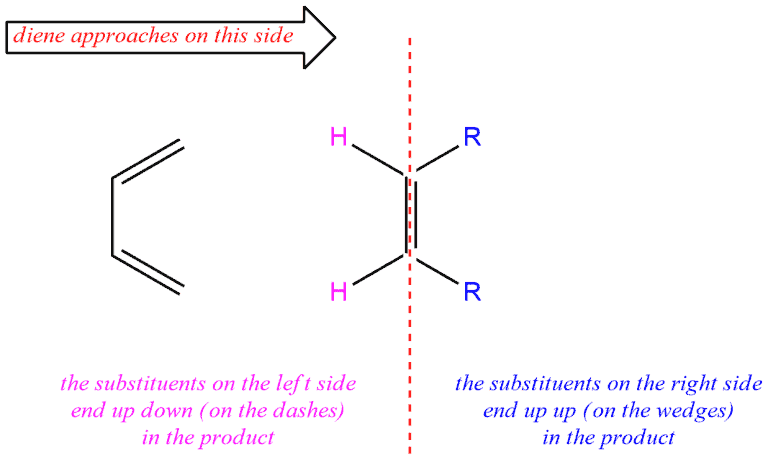
But if think that diene approaches dienophile on the other side, our groups will be on the opposite side of the previous case. So, our R groups will be on the left side, and hydrogens will be on the right side. In the resulting compound, the R groups will be drawn on the dashes, and hydrogens on the wedges.
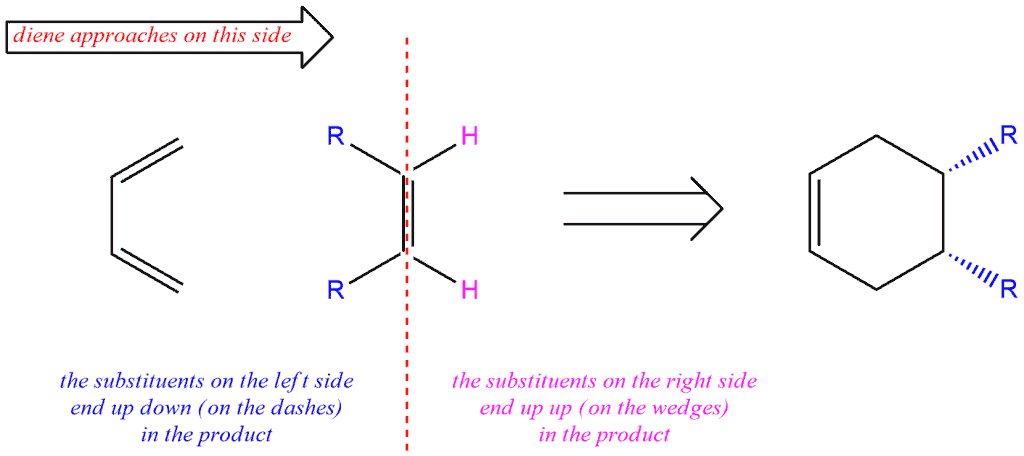
Now, if we have dienophile with two R groups which are trans to each other, in the product these R groups will be up and down, i.e. on a wedge and a dash. The R group which is drawn on the left side of the double bond, in the product will be on a dash, and the R group which is drawn on the right side of the double bond will be on a wedge.
Stereochemistry of diene
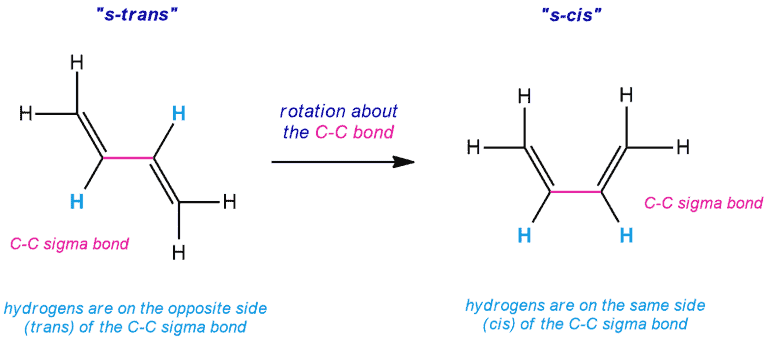
The 1,3-butadiene exists as an equilibrium between the s-cis conformation and the s-trans conformation, and the Diels-Alder reaction only occurs when the diene is in an s-cis conformation. When the compound is in an s-trans conformation, the ends of the diene are too far apart to react with the dienophile.
Similar to dienophiles, the stereochemistry of the diene also is retained. Note that products here contain stereocenters and may be either meso or chiral. And in the following examples, we can see both cases. In the first example, both double bonds are trans in the starting diene. As a product, we get a meso compound. The groups that were on the right side of the diene are now on a wedge, and the ones on the left are on a dash. In the next two reactions, we have the same dienes but in the different perspective. This means that as a product we get these pair of enantiomers.
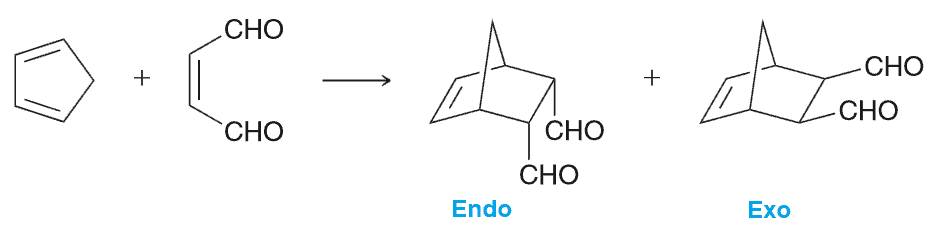
Endo rule
When cyclopentadiene is used as the starting diene, a bridged bicyclic compound is obtained as the product. A bicyclic ring system in which the two rings share non-adjacent carbon atoms is called a bridged ring system. In such a case, we might expect to obtain the following two products:
An electron-withdrawing substituent on one bridge is endo if it is closer to the longer bridge that joins the two carbons common to both rings. The longer bridge has two carbon atoms and the double bond which is formed in this reaction.
An electron-withdrawing substituent is exo if it is closer to the shorter (methylene) bridge that joins the carbons together.
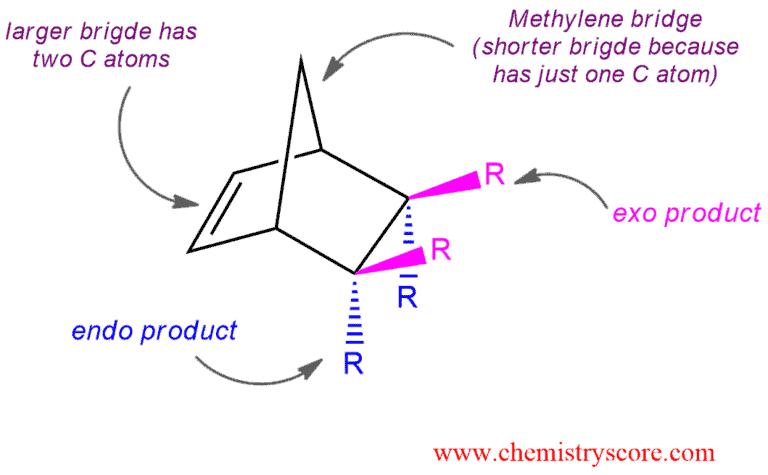
Which of these two products will be obtained? How do we know that?
Using the Endo rule!
When “endo” and “exo” products are possible, the “endo” product is preferred.
The endo product is formed faster than the alternative exo isomer. This occurs even though the exo product is often more stable than its endo counterpart. The preference for endo cycloaddition has its origin in a variety of steric and electronic influences on the transition state of the reaction. Although the endo transition state is only slightly lower in energy, this is sufficient to control the outcome of most Diels-Alder reactions. Mixtures may ensue in the case of highly substituted systems or when several different activating substituents are present.
If you have any questions or would like to share your reviews on the Diels-Alder reaction, then comment down below. I would love to hear what you have to think.
