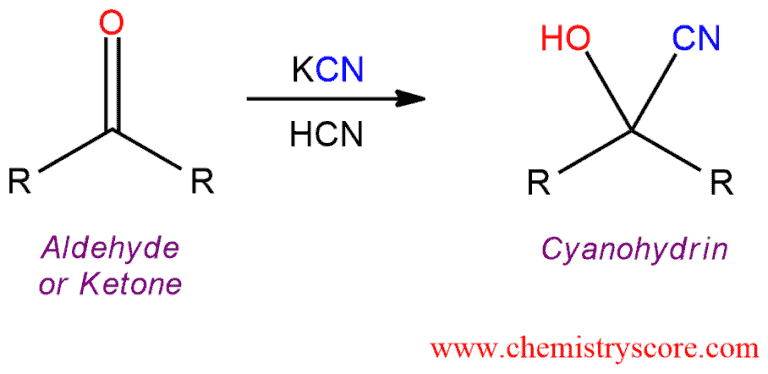
Cyanohydrin formation [CN(–)]
Cyanohydrin formation [CN‾] Definition:
When treated with hydrogen cyanide (HCN), aldehydes and ketones are converted into cyanohydrins which are characterized by the presence of a cyano group and a hydroxyl group connected to the same carbon atom.
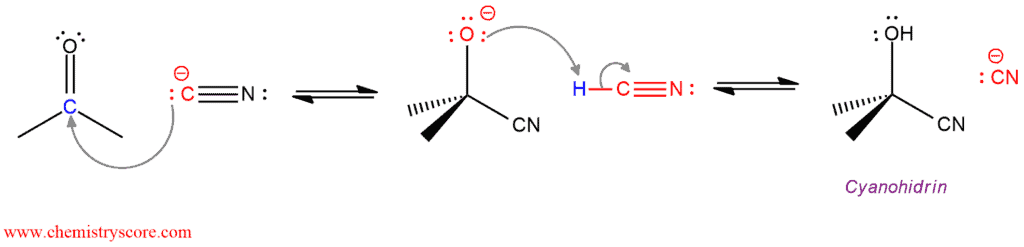
Cyanohydrin formation [CN‾] Explained:
The carbonyl group of aldehydes and ketones can be attacked by carbon-based nucleophiles such as hydrogen cyanide (HCN). Particularly important are carbon nucleophiles because new carbon-carbon bonds can be made in this way. Hydrogen cyanide adds reversibly to the carbonyl group to form cyanohydrins. Cyanohydrins are useful intermediates because the nitrile group can be modified by further reaction.
In the presence of a catalytic amount of base, a small amount of hydrogen is deprotonated to give cyanide ions which catalyze the reaction. This cyanide ion, in the first step, functions as a nucleophile and attacks the carbonyl group. This step is slow and reversible. The negatively charged oxygen of this intermediate abstracts a proton from hydrogen cyanide, regenerating cyanide ion (catalyst) and forming final product cyanohydrin.