Curtius Rearrangement
Curtius Rearrangement Definition:
The Curtius rearrangement is a rearrangement of acyl azides to give isocyanates, expelling the nitrogen gas. The isocyanates can then be used in the other reactions to give products such as ureas and carbamates.
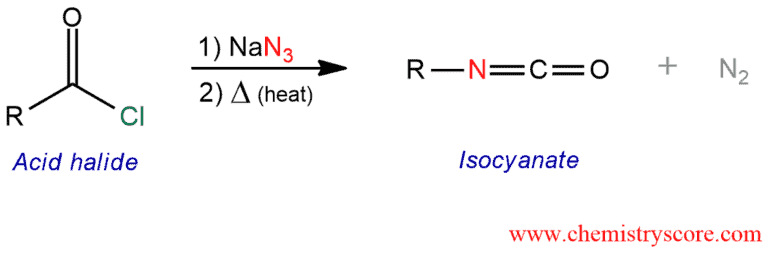
Curtius Rearrangement Explained:
The Curtius rearrangement is similar to the Hofmann rearrangement. But as the starting material, acid chloride is used instead of the amide. And the reaction takes place under neutral conditions instead of the basic conditions.
The mechanism of this reaction consists of an alkyl shift of the R group from the carbonyl carbon to the closest nitrogen with the release of nitrogen gas. Unlike Hofmann rearrangement, where isocyanate is intermediate which cannot be isolated, but here the isocyanate can be isolated and further reacted in the presence of nucleophiles.
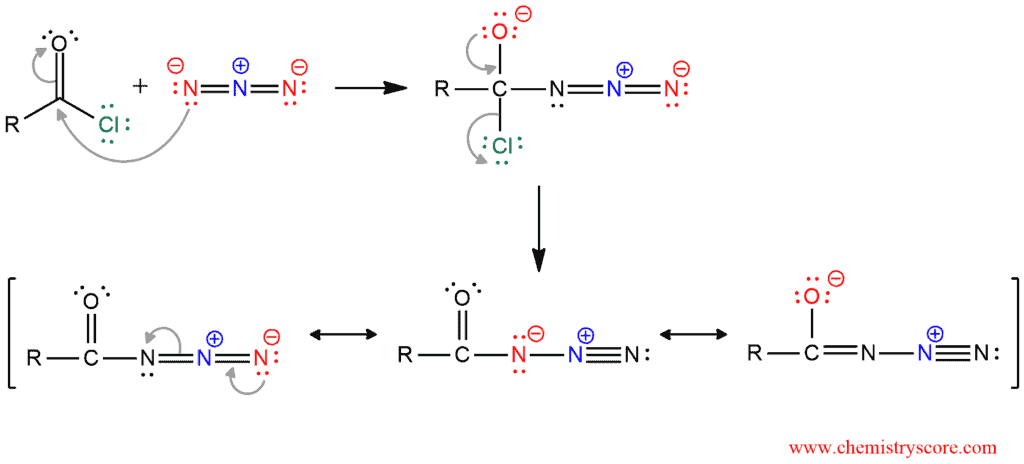
The first step of the mechanism is the formation of acyl azides. When added azide (NaN3) to acid chloride, the tetrahedral intermediate is formed. By reforming the carbonyl bond, the chloride ion as a good leaving group is expelled. The resonance-stabilized ion is formed.
The next step is the thermal decomposition of carboxylic azides to produce isocyanate. The Curtius rearrangement occurs in this step and it is very similar to the last step of Hofmann rearrangement. When carbonyl group is reformed, the loss of the leaving group (nitrogen gas) followed by the migration of the alkyl (R) group from the carbonyl carbon to the closest nitrogen. The product of this rearrangement is an isocyanate.
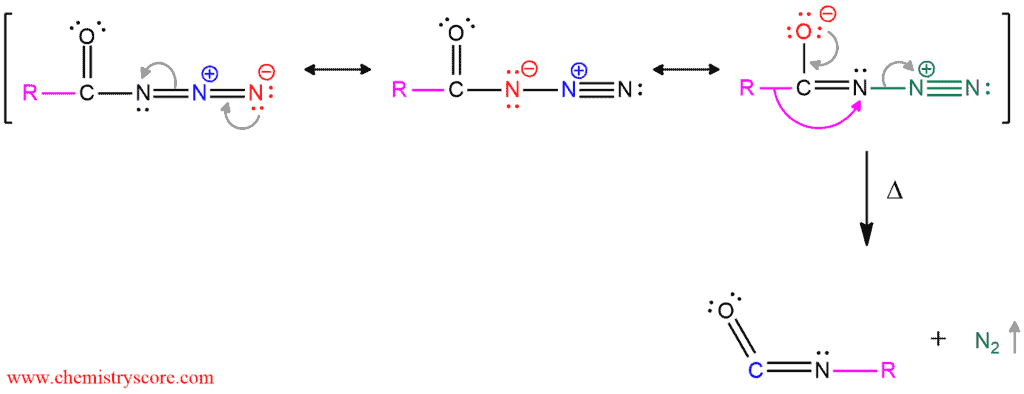
And this reaction can also be completed as the Hofmann rearrangement. This isocyanate can be subject to the reaction with water to the unstable carbamic acid derivative which will undergo spontaneous decarboxylation. Then, this product can be used as versatile starting materials in various reactions and as a monomer for polymerization work and in derivatization of biomolecules.