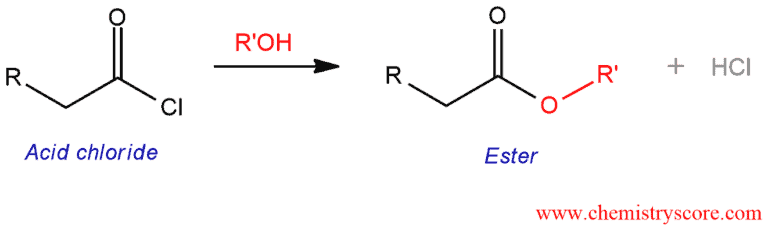
Conversion to esters [ROH]
Conversion to esters [ROH] Definition:
Acid chlorides are converted to esters when treated with alcohols. This reaction called alcoholysis.
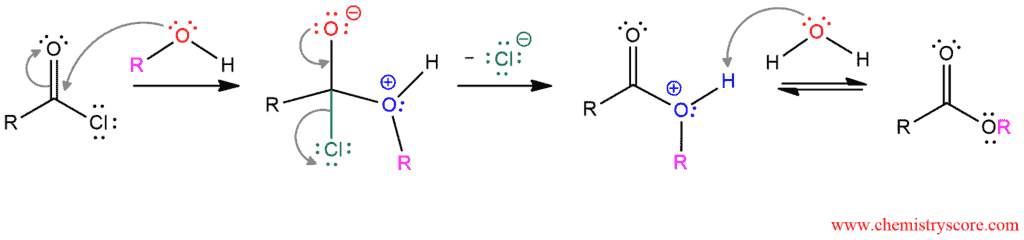
Conversion to esters [ROH] Explained:
This reaction is analogous to the conversion of acyl chlorides with water and is a very useful way of producing esters. Because acyl chlorides are easily obtained from the corresponding carboxylic acids. Here, the hydrogen chloride is also an undesired by-product that can be neutralized with a base such as an alkali metal hydroxide, pyridine, or a tertiary amine.
The mechanism is also directly analogous to the mechanism of hydrolysis of an acyl chloride. It consists of three steps, and in the first, alcohol function as a nucleophile and attacks the carbonyl carbon. In the second step, the carbonyl group is re-formed by expelling a chloride ion which is a good leaving group. And in the last step, the positive charge of the oxygen is removed by protonation and the ester is formed.