Chlorohydrin formation [Cl2/H2O]
Chlorohydrin Formation Definition:
The chlorohydrin formation reaction involves adding chlorine and water to a double bond and creating a chlorohydrin (chloro=chlorine, hydrin=hydro, water/H2O).
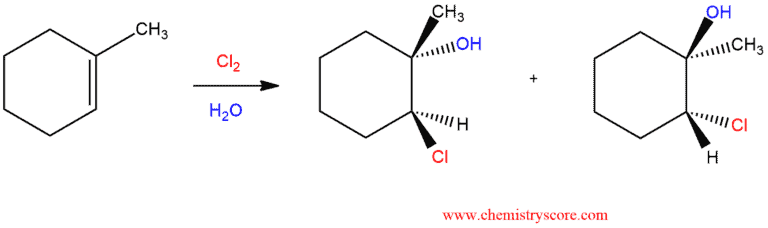
Chlorohydrin Formation Explained:
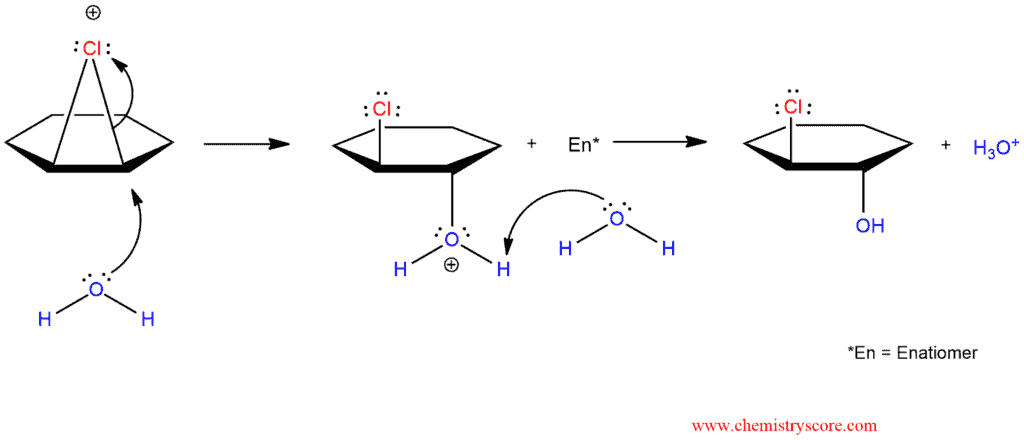
When chlorination occurs in a non-nucleophilic solvent, such as CHCl3 or CCl4, the result is the addition of Cl2 across the π bond. However, when the reaction is performed in the presence of water, the chloronium ion that is initially formed can be captured by a water molecule, rather than chloride:
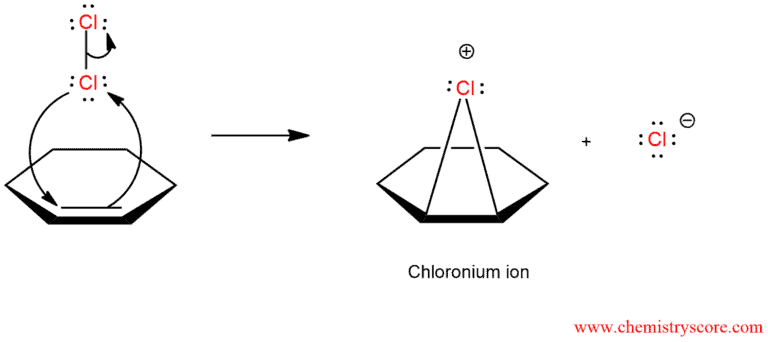
As we can see, chlorohydrin formation begins like chlorination but in the second step, chloride is not added to chloronium ion because it is surrounded by water and unavailable for the reaction.
Water is solvent and molecules of water are good nucleophiles which will react with chloronium ion. A molecule of water will add to more substituted carbon atom because it is more capable of stabilizing the partial positive charge in the transition state. The proposed mechanism is therefore consistent with the observed regioselectivity of halohydrin formation.