Chlorination [Cl2 plus catalyst]
Chlorination [Cl2 plus catalyst] Definition:
Treatment of an aromatic (such as benzene) with chlorine (Cl2) and a Lewis base such as AlCl3 or FeCl3 leads to the formation of the chlorinated aromatic by electrophilic aromatic substitution.
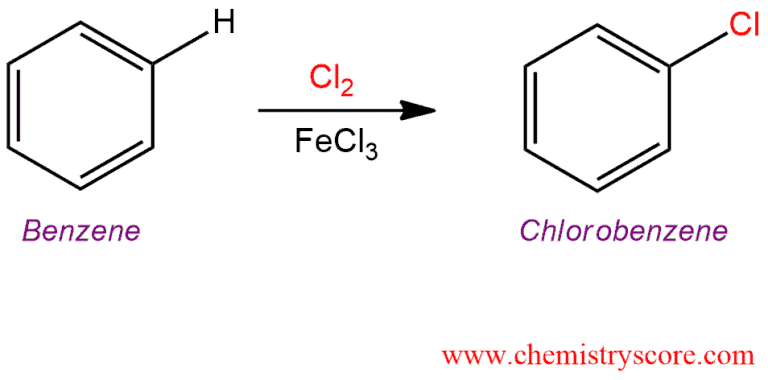
Chlorination [Cl2 plus catalyst] Explained:
This reaction is similar to catalytic aromatic bromination. Benzene does undergo electrophilic aromatic substitution with chlorine and aluminum chloride as a catalyst to produce chlorobenzene.
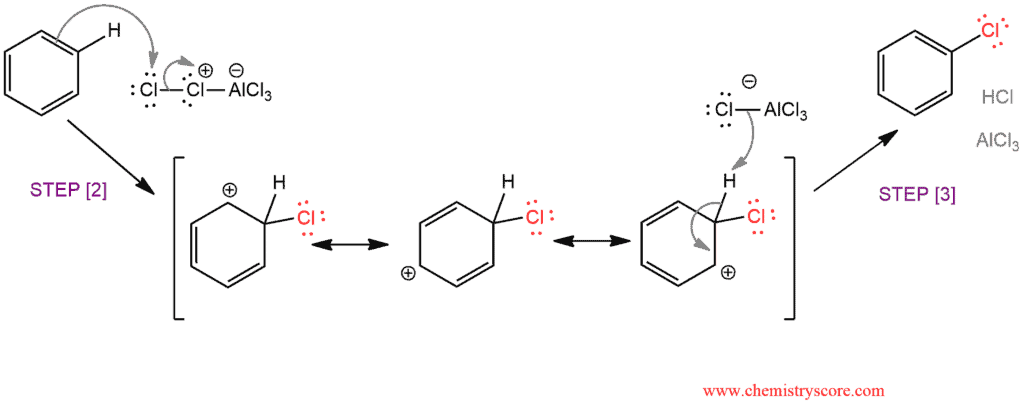
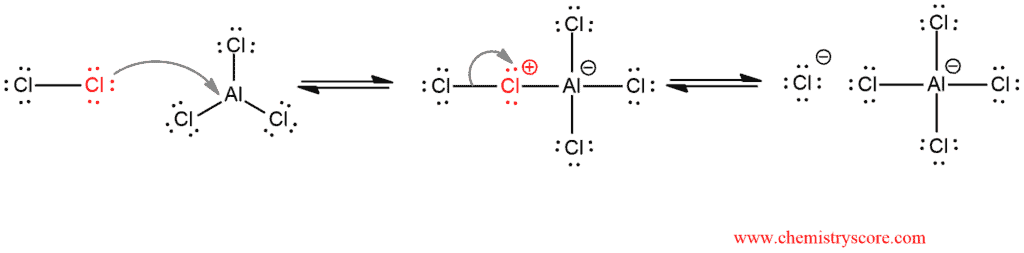
Chlorine reacts with AlCl3 to form a complex in which imparting electrophilic character to the chlorine atom [1]. This electrophilic chlorine atom reacts in the next step [2], the nucleophilic attack of benzene. Sigma complex, or arenium ion, is also formed. The reaction ends [3] by deprotonation of sigma complex and, thereby, restoring aromaticity and regenerating the Lewis acid (AlCl3).
Activation of chlorine by the Lewis acid AlCl3
Electrophilic attack on Benzene by Activated Chlorine and Chlorobenzene formation