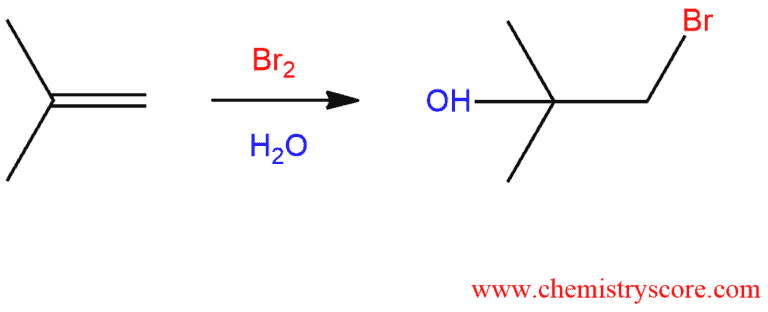
Bromohydrin formation [Br2/H2O]
Bromohydrin Formation Definition:
Bromonydrin formation is addition reaction of Br and OH across the alkene and the product is called a bromohydrin (bromo=bromine, hydrin=hydro, water/H2O).
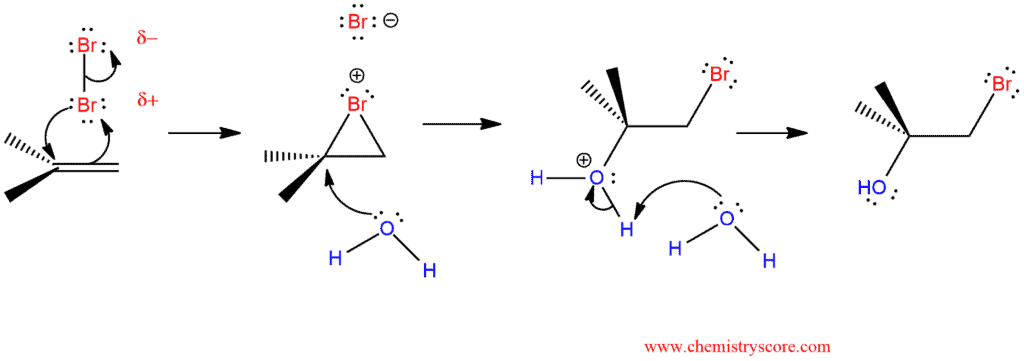
Bromohydrin Formation Explained:
Lewis acid like the bromine is able to bond to the alkene π electrons, and the resulting complex rearranges or is attacked by nucleophiles to give addition products.
The intermediate bromonium ion is a high-energy intermediate and will react with any nucleophile that it encounters. When water is the solvent, it is more likely that the bromonium ion will be captured by a water molecule before having a chance to react with a bromide ion. So water will add to more substitute carbon by the SN2 mechanism. This leads to an anti-product and this means that will the bromine and the hydroxyl group will be on the opposite sides.