Allylic bromination [NBS]
Allylic Bromination Definition:
When treated with N-bromosuccinimide (NBS) and light (hν) alkyl groups adjacent to the alkenes will be converted into alkyl bromides.
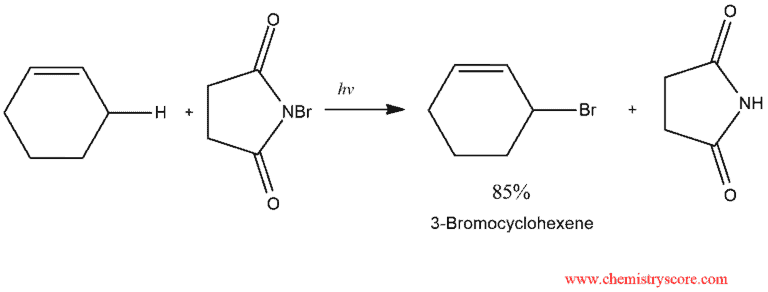
Allylic Bromination Explained:
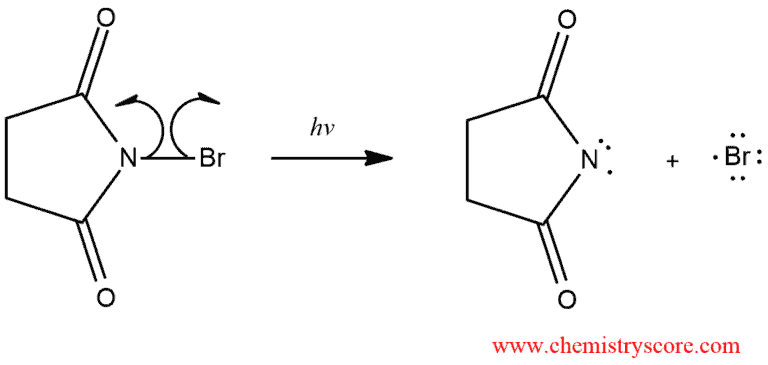
Under radical conditions, alkanes containing allylic hydrogens enter into allylic halogenation. A particularly good reagent for allylic bromination is N-bromosuccinimide (NBS). The bromine reacts with the alkene by radical chain mechanism. The process is initiated by light that causes dissociation of Br2 into bromine atom.
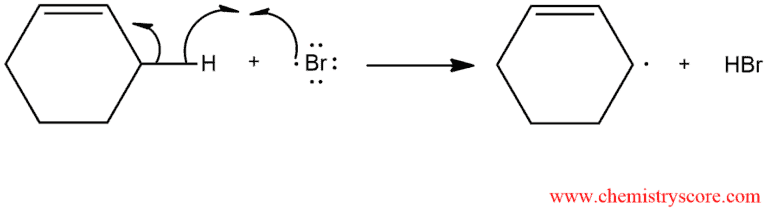
Propagation of the chain involves abstraction of weakly bound allylic hydrogen by bromine atom.
The HBr produced in this step then react with NBS in an ionic reaction that produces Br2. This Br2 is then used in the second propagation step to form the product:
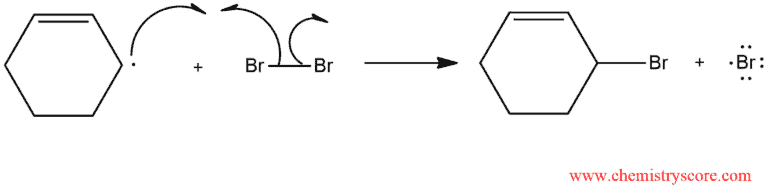
Throughout the process, concentrations of HBr and Br2 are kept at a minimum. Under these conditions, the ionic addition of Br2 does not successfully compete with radical bromination.
The resonance-stabilized radical may then react Br2 and be undergoing to halogen abstraction at any location. When placing the bromine in the position of the non-contaminated electron in each resonant structure, giving the following products:
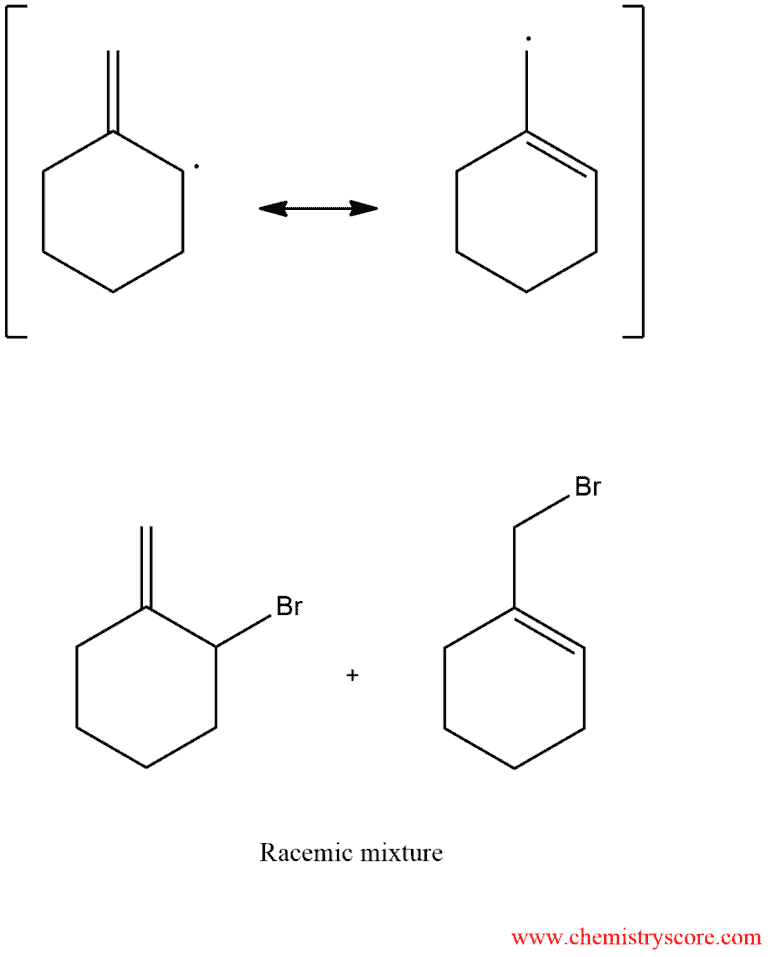
Thus, alkenes that form unsymmetric allylic radicals can give mixtures of the products upon treatment with NBS.