Aldol Condensation
Aldol Condensation Definition:
When heated in acidic or basic conditions, the product of an aldol addition reaction will undergo elimination to produce unsaturation between the α and β positions.
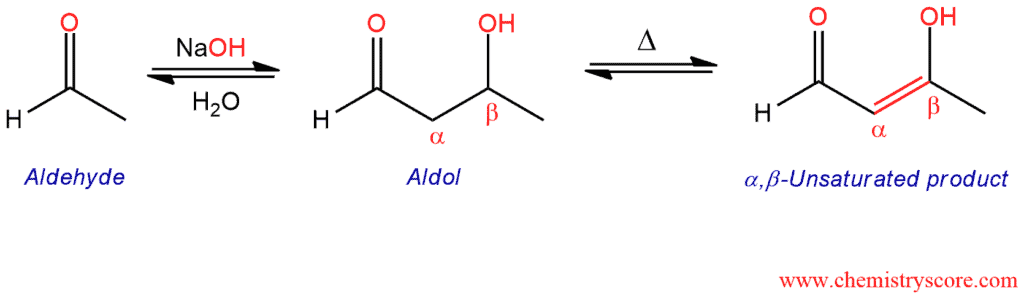
Aldol Condensation Explained:
The aldol condensation is a reaction between an enolate (or enol) and an aldehyde or ketone that leads to the formation of a new carbon-carbon double bond. The reaction is called a “condensation” because two molecules undergo addition accompanied by the loss of a small molecule such as water in this case.
An aldol condensation has two parts:
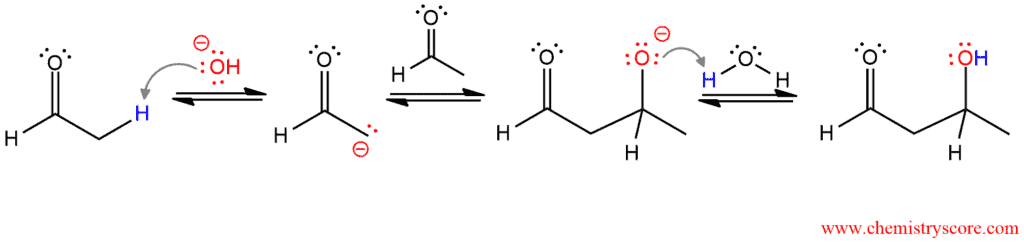
PART 1: This is an aldol addition reaction, which consists of three steps. The hydroxide ion removes the proton from the aldehyde in α position and generates the enolate. This enolate acts as a nucleophile and attacks an aldehyde, and the alkoxide ion is formed. In the last step, the resulting alkoxide ion is protonated by water to give β-hydroxy aldehyde.
PART 2: The elimination of water – In the presence of strong base, the dehydration reaction occurs. First, the α position is deprotonated to form an enolate, and then the hydroxide is ejected to afford the product.
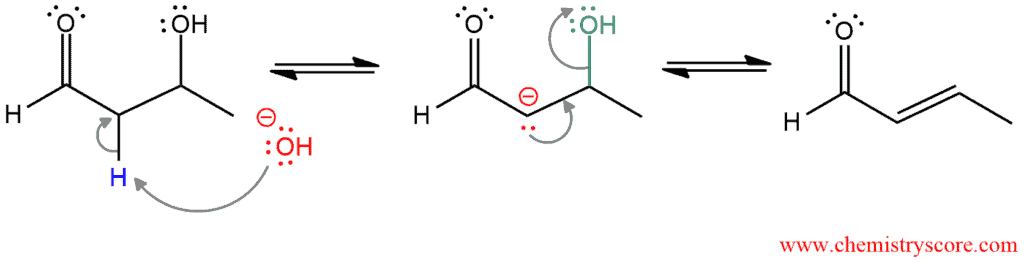
In some cases, the aldol addition product cannot be isolated. Even at the moderate temperatures, only the condensation product is obtained. But sometimes where the aldol addition product can be isolated (by performing the reaction at low temperature), the yields for condensation reaction are often much greater than the yields for addition reaction.
The aldol condensation is a reaction that occurs between two identical compounds. But this reaction may occur between two different carbonyl compounds and is called a crossed aldol, or mixed aldol, reaction. And with diketones (have two carbonyl group in one molecule), and intramolecular aldol condensation occurs.