Addition of Gilman to acyl halides
Addition of Gilman to acyl halides Definition:
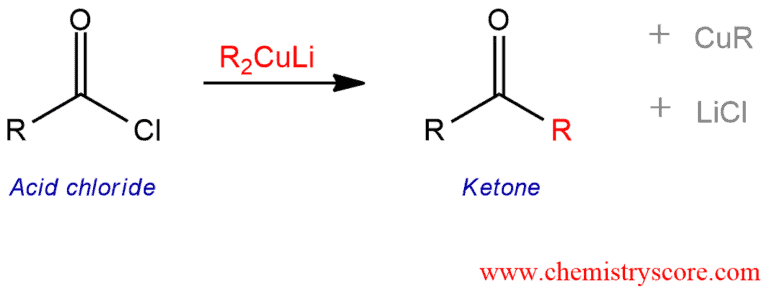
Addition of organocuprates (Gilman reagents) to acid chlorides results in ketones.
Addition of Gilman to acyl halides Explained:
Organometallic reagents may attack the carbonyl group of acyl halides to give the corresponding ketones. Organolithiums and Grignards are relatively unselective and products from this reactions may further react. Ketone formation is best achieved by using organocuprates (Gilman reagents), which are more selective than organolithium and Grignard reagents and do not add to the product ketone. This reaction results in a nucleophilic substitution of an alkyl group for the leaving group (chloride), forming one new carbon-carbon bond.
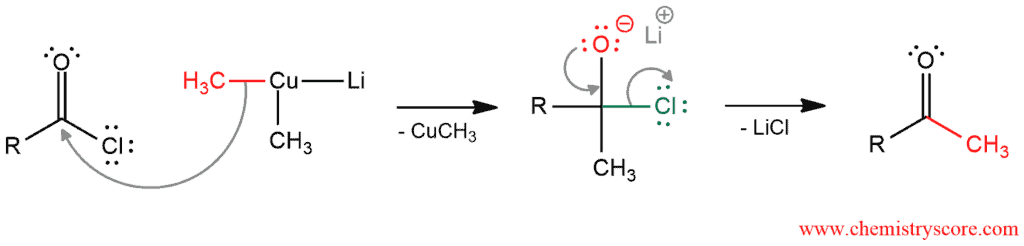
Mechanism of Addition of Gilman to Acyl Halides
This mechanism consists of two steps. In the first step, one of the methyl groups of organocuprate function as a nucleophile and attacks carbonyl carbon (Remeber: whenever a carbon is attached to a metal it has a negative charge). π bond will break and in the next step, this bond will re-form by expelling a good leaving group (chloride ion). And the final ketone will be formed.