All animal cells have a lipid bilayer membrane that interacts in the environment that it is in. This means that various proteins come and go, resulting in interactions that characterize cellular function.
Some proteins are components of lipid bilayers, while others come and go as needed.
In this article, we’ll go over the basics of membrane proteins.
Types of Proteins:
Peripheral Proteins:
Peripheral proteins (or extrinsic) proteins bind loosely to the lipid bilayer surface and interact with the polar head groups.
The types of interactions involved could be:
- Hydrogen bonds between protein and polar head groups
- Electrostatic interactions
Note: the interactions between peripheral proteins is so loose that it is easy to disrupt the interaction.
Integral Proteins:
Integral proteins or (intrinsic) proteins are inserted into the bilayer and can interact with both the polar head groups and the hydorphobic tail gorups and surface of a membrane (amphiphillic)
Much stronger attachments than peripheral protein.
Transmembrane Proteins:
A type of integral protein that spans across the entire lipid bilayer.
Typically you might have a protein that has hydrophillic amino acids on the outside while the more nonpolar amino acids span the membrane.
Common transmembrane structures:
- Bacteriorhodopsin: an example of a transmembrane protein with an alpha-helical structure (note: alpha-helical structure is the most ideal for transmembrane because the polar groups are already tied up in the structure of the backbone, while sidechains are all hydrophobic.
- Porins or beta barrels shape: an example of a transmembrane protein with a beta barrel shape that has polar backbone groups in H-bonds, while the side chains depends on the orientation. If the orientation of side-chains is out, then they will interact with fatty acid tails. If side-chains point in, then it could be either hydrophobic or hydrophilic depending on specific function.
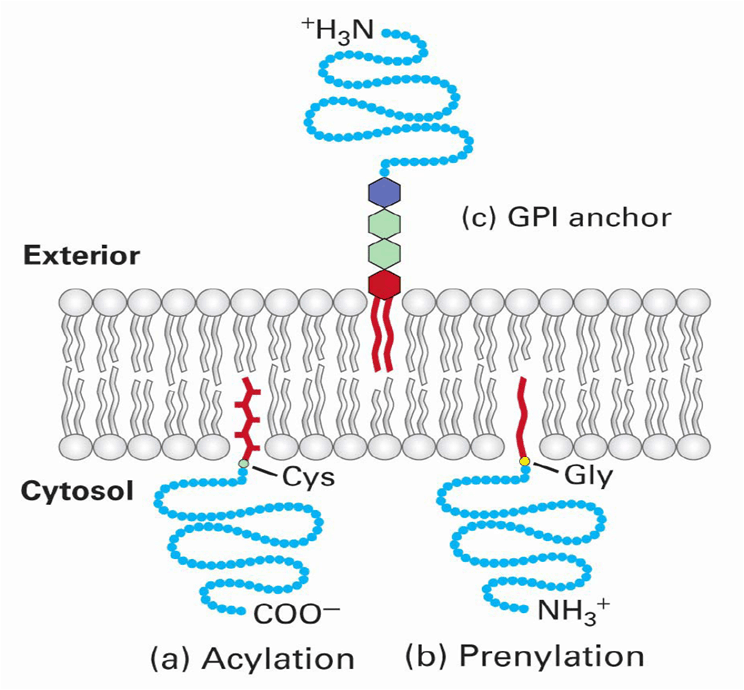
Lipid-Linked Proteins:
Lipid-linked proteins are found are the surface of a bilayer by a covalent anchor to the protein.
Few different types of covalent linkages:
- Prenylated
- Fatty acid acylated
- Glycosylphospatidylinositol-linked (GPI-linked)
Prenylation:
In prenylation, the protein is attached to hydrophobic groups built from isoprene units and are attached via thioether linkage to Cys at C-X-X-Y recognition sequences. (X= irrelevant amino acid; Y = amino acid that determines type of prenylation .
Fatty Acid Acylation
In fatty acid acylation, a fatty acid is linked to the protein.
- Myristoylation
- In the case of myristoylation, there is an N-linked to N-terminal glycine via amide linkage
- Palmitoylation
- Thioester to a specific cystein
GPI-Linked
In Glycosylphosphatidylinositol-linkage you simply have the GPI group link to your protein.