Organic chemistry is reduced to consideration of the reactions and mechanisms of organic compounds. Learning how to name organic compounds is the foundation upon which your entire organic chemistry knowledge will be based.
The nomenclature of the linear molecules is simple, but when these molecules begin to branch, the naming of organic compounds becomes very complex.
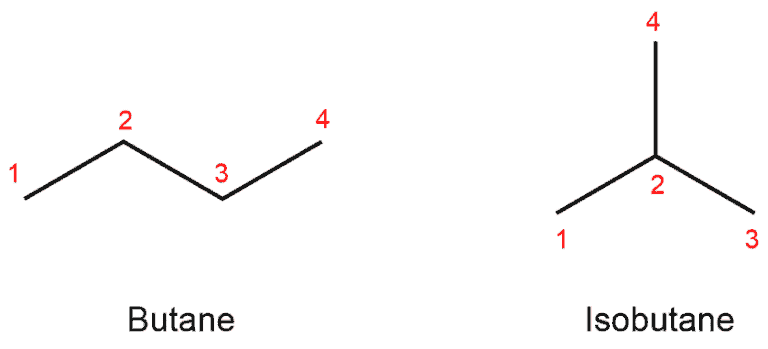
The first situation we encounter is branched alkyl substituents on alkanes. So let’s consider the simplest example, and it is butane (alkane) or butyl (alkyl group).
Butane has four carbon and thus two possible structures. These are the so-called structural isomers. One is the 4-carbon straight chain which is call n-butane (“n” like normal), and the other is the 3-carbon chain with a methyl group on C2. It is called 2-methyl propane, or isobutane.
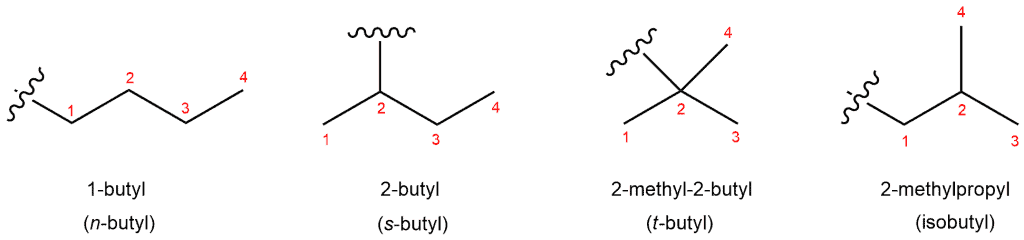
When we remove a hydrogen from butane we get butyl group. There are four different types of “butyls”, and they all have their own name:
1-butyl (n-butyl where “n” stands for “normal”)
2-butyl (s-butyl where “s” stands for “secondary”)
2-methyl-1-propyl (isobutyl)
2-methyl-2-propyl (t-butyl)
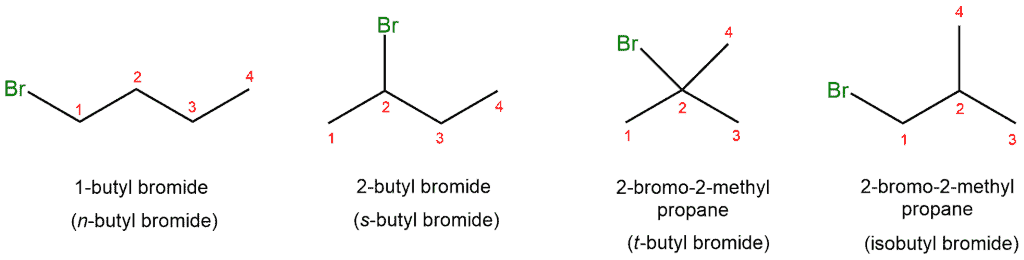
For example, chlorination of butane would result in:
Therefore, giving a name to a branched molecule will follow the following items:
- Identify the parent chain or the largest chain in the molecule (marked green)
- Numerization of the parent chain – the numbering is done on one side where the branching of the molecules is closer (1-10)
- then identify the attached group (marked blue)
- and number the attached group (1-3)
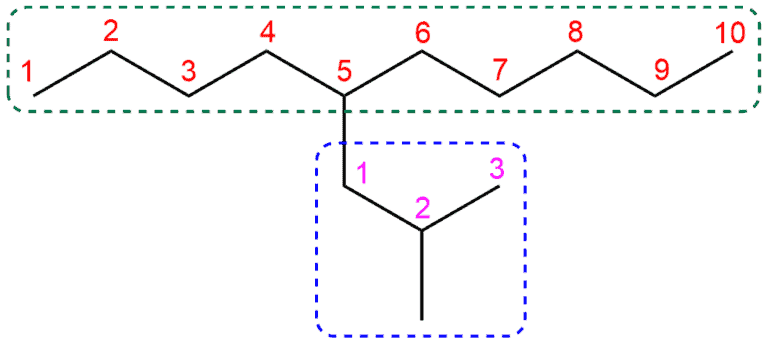
Thus, the name of our compound in this example is 5-(2–methylpropyl)decane or 5–isobutyldecane.