Both hemoglobin and myoglobin are present in our bodies to help with blood transportation.
However, both has distinct functions and structural properties that allow them to carry out different tasks in our body.
First, let’s begin with hemoglobin:
Structure of Hemoglobin:
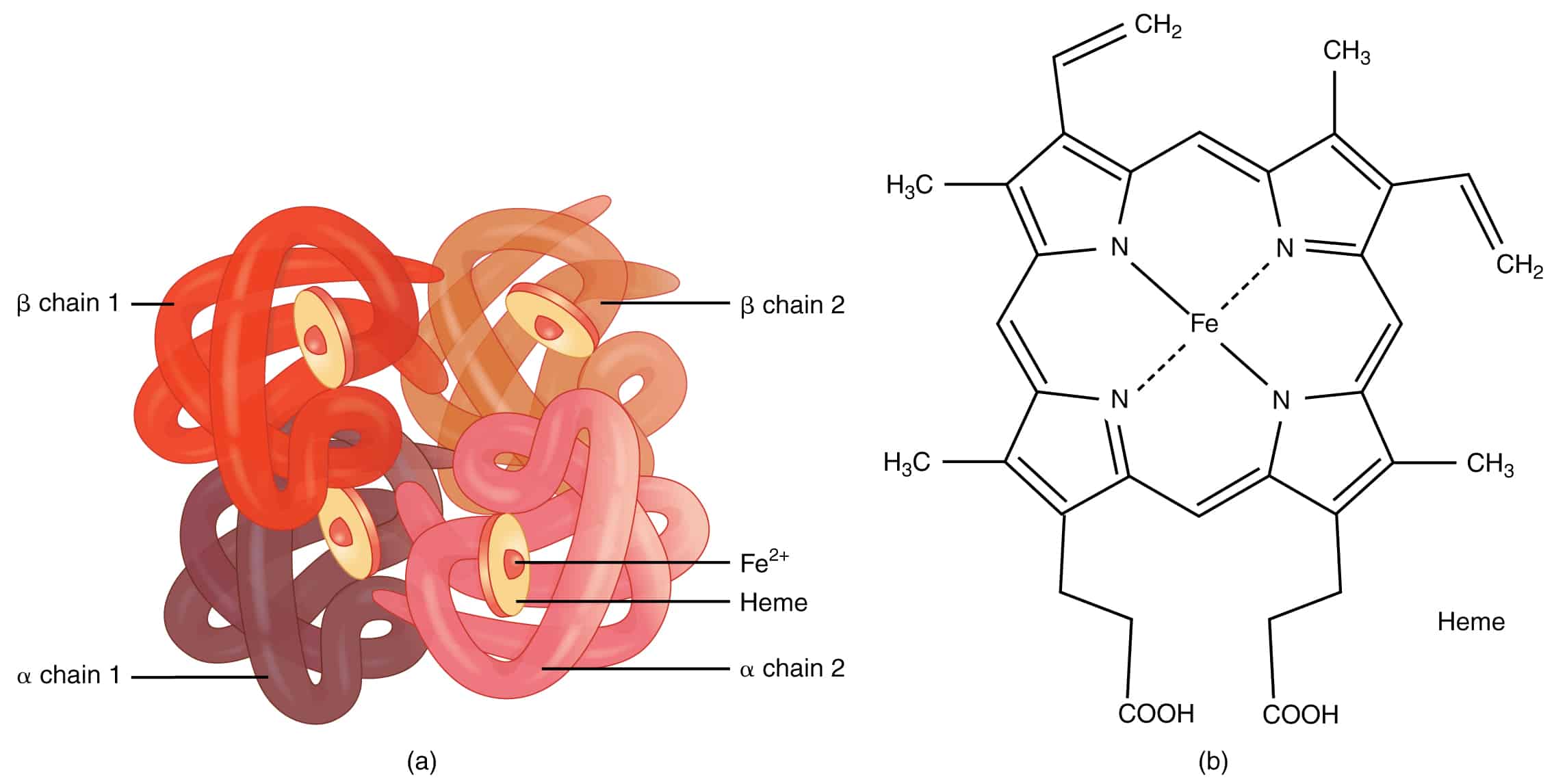
Structural Properties of Hemoglobin:
- Hemoglobin is a tetramer meaning there are four separate chains held together by iron (Fe2+)
- There are two beta chains and two alpha chains (a2b2)
- Dimensions of hemoglobin: 64 X 55 X 50 angstroms
- Oxygen binding alters the structure of hemoglobin, therefore deoxyhemoglobin and oxyhemoglobin are noticeably different.
- The structure has symmetry a1b1 (side #1) and a2b2 (side #2)
- a1b1 has 35 residues while a1b2 has 18 residues
- When oxygen binds to hemoglobin, the oxygenation results in one ab dimer to shift 15 degrees with respect to the other ab dimer. This is important as this structural rearrangement defines hemoglobins oxygen-binding behavior.
Hemoglobin Oxygen Binding Cooperativity:
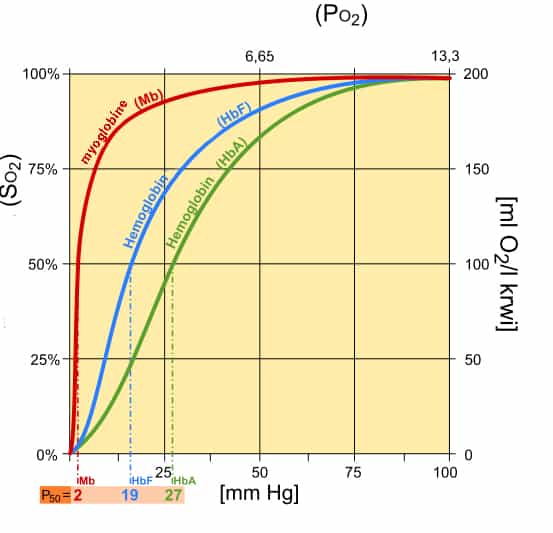
Hemoglobin (HbA) and fetal hemoglobin (HbF) resemble a sigmoidal curve when it comes to oxygen binding affinity.
This is because hemoglobin has something called cooperative interaction between binding sites.
Cooperative binding = binding of a ligand to one site increases affinity for a ligand at another binding site.
Since hemoglobin is a tetramer, cooperative binding plays an important role.
The p50 (pressure required for hemoglobin to be 50% bound to oxygen) is much higher as opposed to myoglobin.
This allows for the blood to deliver more oxygen to the tissues than myoglobin since the binding affinity is less than that of myoglobin.
Hemoglobin binding to oxygen can be defined using the Hill Equation:
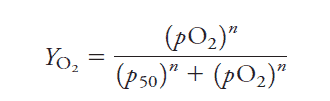
The ‘n’ refers to the number of binding spots in hemoglobin. Since hemoglobin is a tetramer, oxygen can bind to four locations.
If n = 1, O2 binding is not cooperative
If n > 1, O2 binding is positively cooperative meaning O2 binding increases affinity for further O2 binding.
If n < 1, O2 binding is negatively cooperative because O2 binding would reduce affinity of hemoglobin for subsequent O2 binding.
Hemoglobin Conformations: R State and T State
The cooperativity of oxygen binding to hemoglobin results from the effect of ligand-binding state of one heme group on the ligand-binding affinity of another.
What this means is that if oxygen binds to one of the four heme groups of hemoglobin, the affinity of another heme group ch
Hemoglobin vs. Myoglobin:
| Hemoglobin | Myoglobin | |
| Structure | Tetramer (Oxygen can bind to four locations) | Monomer |
| Transport | Oxygen transport in blood | Oxygen transport to tissues |
| Oxygen binding | Sigmoidal | hyperbolic |
Structure of the Heme Group:
The heme group is comprised of the following key features:
- Heterocyclic porphyrin ring consisting of 4 pyrrole groups
- The nitrogen’s of the pyrrole groups, a histidine, and oxygen coordinate the Fe(II) ion located in the center
- Oxygen binding is reversible
- red blood = oxygenated
- purple/blue blood = deoxygenated
- Heme group can coordinate CO (carbon monoxide binding as well)
