Heck Reaction
Heck Reaction Definition:
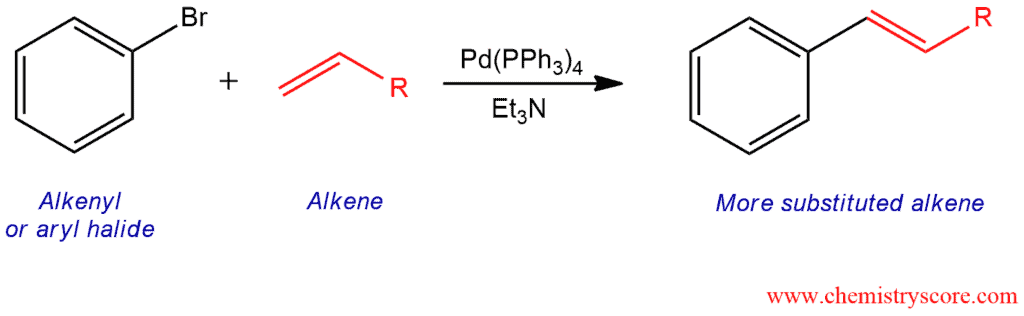
The Heck reaction is a coupling reaction of a vinyl or aryl halide with an alkene in the presence of a palladium catalyst which forms a more highly substituted alkene with a new carbon-carbon bond.
Heck reaction Explained:
The Heck reaction is the type of coupling reaction where vinylic (alkenyl) or aryl halide, R’-X (X=Cl, Br, I) react with an alkene but it needs a palladium catalyst. As a catalyst is usually used tetrakis(triphenylphosphine)palladium, Pd(PPh3)4, and the reaction is carried out in the presence of a base such as triethylamine. This reaction is a substitution reaction in which one hydrogen atom of the starting alkene is replaced by the R’ group of the alkenyl or aryl halide.
Mechanism of Heck reaction
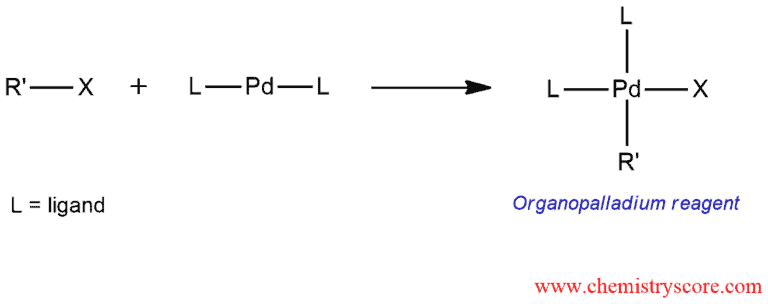
The first step is the formation of an organopalladium compound by oxidative addition. Alkenyl or aryl halide reacts with a palladium catalyst and called oxidative addition because two groups are added to the palladium.
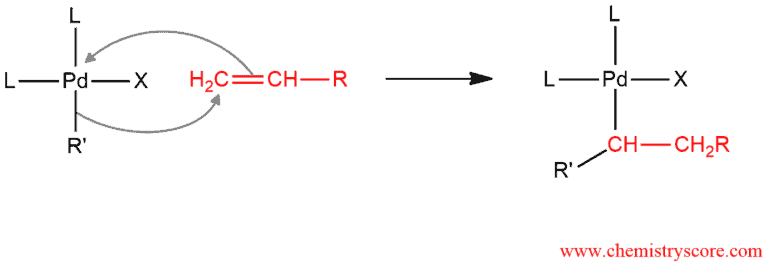
The second step is the addition of alkenyl or aryl group (R‘) and palladium to the double bond of an alkene. R’ group attacks sp² hybridized carbon atom of an alkene. π bond breaks and attack palladium to form a neutral compound.
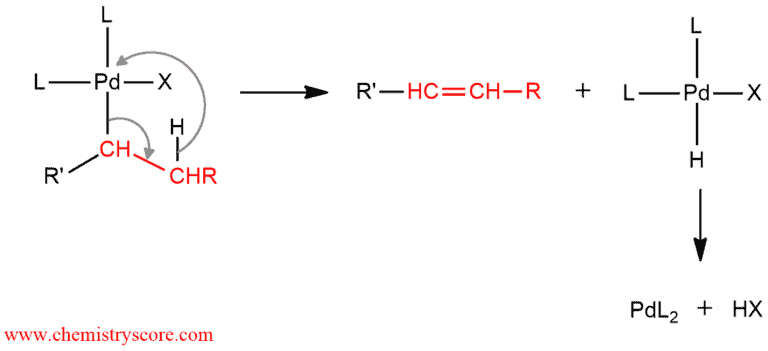
The third and fourth steps are two successive eliminations. First is an syn-intramolecular β-elimination. Palladium has a really large group which would cause a lot of steric issues. A hydride shift over onto the palladium and electrons from palladium-carbon bond (of the large group) will reform the double bond of the starting alkene. Thus, four substituted groups on the palladium are obtained and the more substituted alkene is formed. Because the final product is obtained the reaction may be considered completed. But actually, there is the last step where the palladium catalyst is regenerated. By a reductive elimination of HX, the starting catalyst is obtained.