Sp3-Hybridized Orbitals Definition:
An sp3 hybridized orbital is an atomic orbital achieved by the interference of one s orbital with three p orbitals. This gives rise to four degenerate hybridized atomic orbitals.
Sp3-Hybridized Orbitals Explained:
Example – Carbon atom forming 4 bonds
Orbital diagram for carbon in single bonded molecules:-
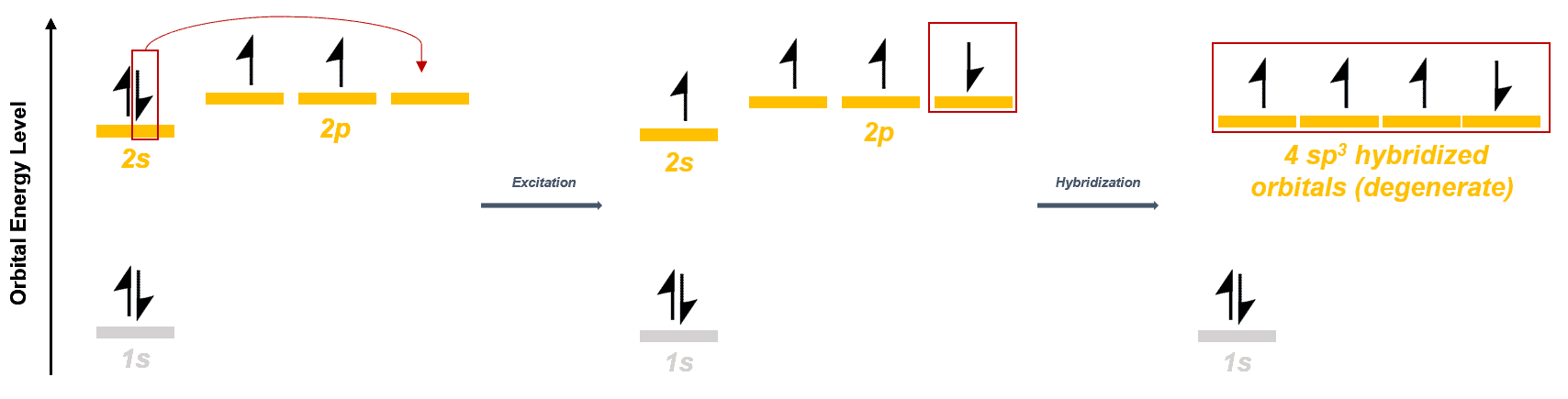
The electronic configuration of carbon tells us that carbon has only 2 unpaired electrons. Hence, to form 4 bonds, the carbon atom promotes one electron from the 2s orbital to the empty 2pz orbital. Now all orbitals are half filled.
However, to explain the molecular structure of simple single bonded molecules like methane, ethane, propane etc, we assume that the orbitals undergo hybridization to give rise to 4 degenerate orbitals (only one shown in the diagram below) which form the 4 bonds. Since one s orbital interacts with 3 p orbitals, the hybridized orbital has 25% s character and 75% p character.