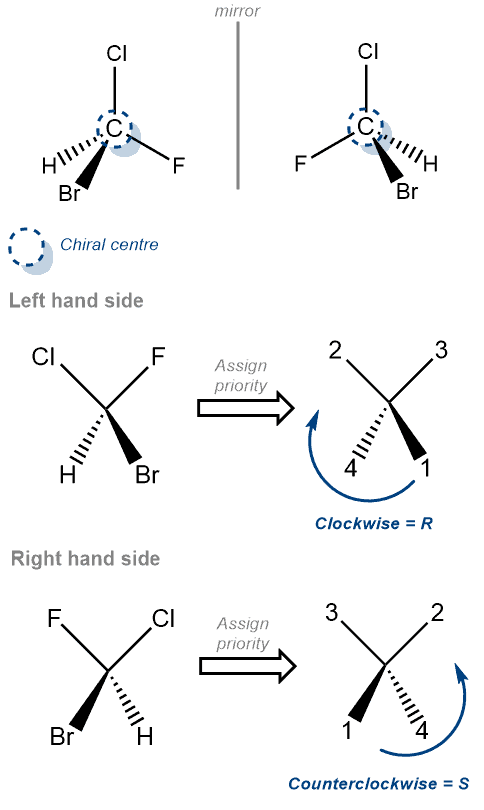
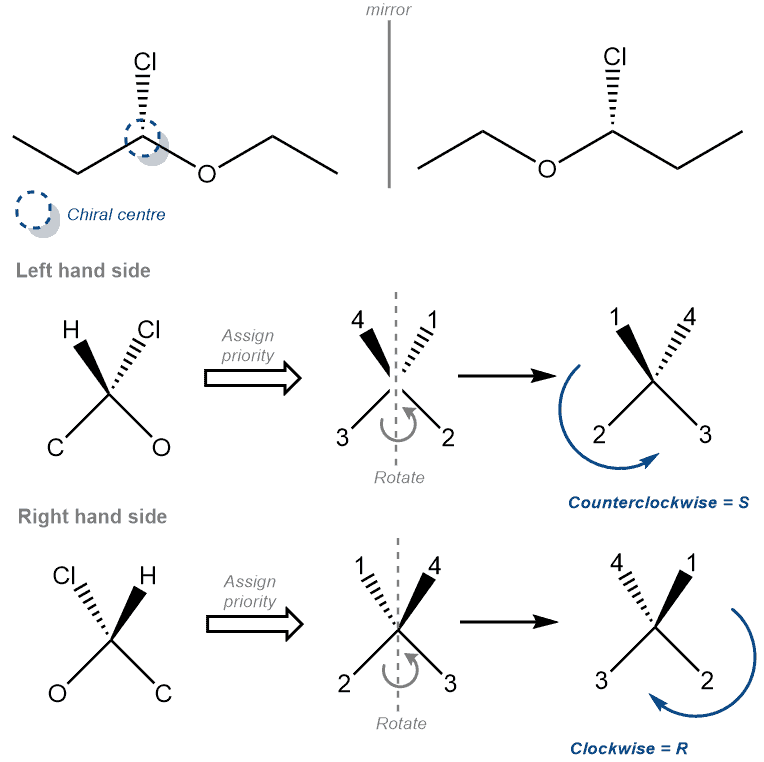
S Definition:
‘S’ is the term used to designate the configuration – counterclockwise – of a chirality center in a molecule.
R Explained:
Chirality talks about molecules that are not superimposable on its mirror image. .The molecule and its mirror image are called enantiomers. As such, to determine or designate each enantiomer we use the ‘R’ and ‘S’ configuration. The ‘S’ configuration represents the molecule which has its 4 groups arranged in a counter-clockwise manner around the chirality center.
To determine the configuration we use the following rules –
- Identify each of the 4 groups and assign a property number to it. (The highest atomic number group gets the highest priority. Therefore, hydrogen is always 4. Example, priority of Cl>O>C>H)
- Rotate the molecule such that group 4 is on a dash bond. i.e. behind the page
- Identify the sequence of priority 1-2-3. Clockwise or Counterclockwise?
Examples –