Pi (p) Bond Definition:
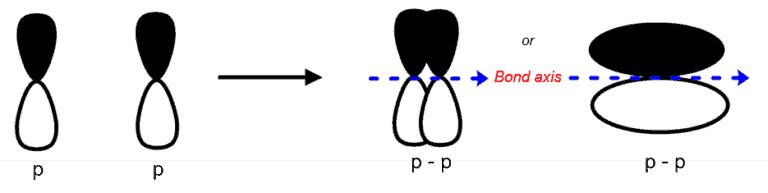
The Pi (p or Π) bond is a covalent bond formed by the lateral overlap between two atomic orbitals.
Pi (p) Bond Explained:
A sigma bond is thus, a bond wherein the maximum density of the electron cloud is perpendicular with respect the axis connecting the centers of the two participating atoms. It represents a weaker (with respect to sigma bonds) overlap and can be thought of as a lateral collision of the orbitals.
Note :
- All double and triple bonds have one and two pi bonds respectively.
- Pi bonds are weaker than sigma bonds.
- Pi bonds do not arise from s orbitals overlaps but arises from the overlap between, p, and d orbitals, like p – p orbitals.
Examples –