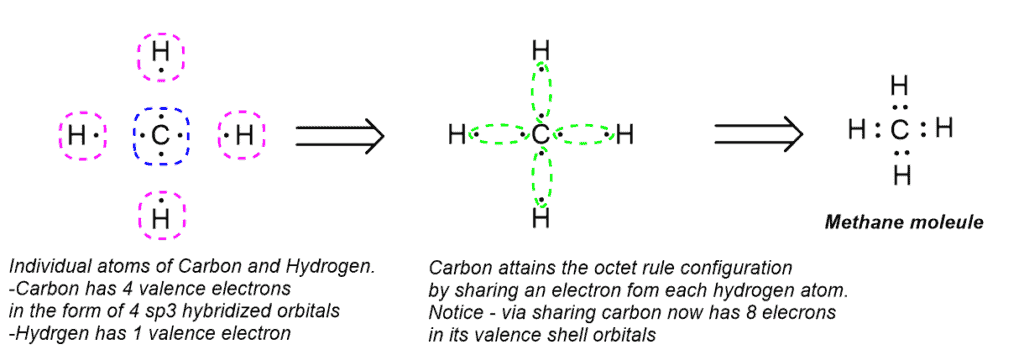
Octet Rule Definition:
The Octet Rule states that elements tend to achieve the electron configuration of the nearest noble gas, which they do by gaining, losing or sharing electrons.
Octet Rule Explained:
The Lewis theory observes that an atom is most stable when its outermost shell or valence shell is completely filled. i.e. it contains 8 electrons for all main group elements. When atoms have less than 8, they tend to react (by giving up, accepting or sharing electrons) to form more stable compounds and attain the completely filled electron configuration.
Also important to note, is that the octet rule considers only the s and p orbitals of the valence shell and not the d and f orbitals. Hence, for all main group elements, the octet corresponds to an electronic configuration ending with s2 p6.
To identify the number of electrons needed to achieve the octet, we should look at the periodic table where elements are grouped by valency (or the number of electrons in the valence shell. Note: Electrons in inner shells, called core electrons, do not participate in chemical bonding. Ony electrons in the outermost shell/valence shell, called valence electrons, participate in chemical bonding. Carbon, for example, has two core electrons and four valence electrons.)
Periodic table groups and associated valency –
Group 1 = +1 Group 2 = +2
Group 13 = +3 Group 14 = +4/-4 Group 15 = -3 Group 16 = -3 Group 17 = -1 Group 18 = 0
Example –