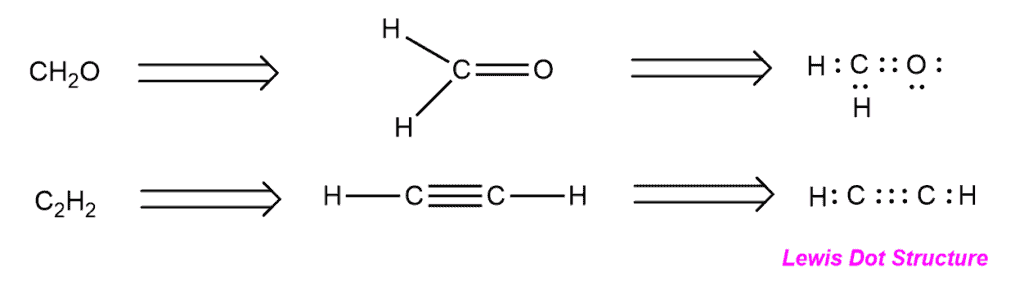
Lewis Structures Definition:
The Lewis Structure or the Lewis Dot Structure is a method of representing molecules where the individual atoms are represented by their periodic symbol (H for Hydrogen, C for Carbon, etc.) around which the number of valence electrons for that atom is drawn.
Lewis Structures Explained:
The Lewis structure is an easy way to interpret the number of bonds an atom will form, its lone pair electrons, and predicting the number of electrons it will need/give up to conform to the octet rule. For Example, in drawing the lewis dot structure for Carbon we draw 4 valence electrons around it and thus know that it will need 4 more electrons to conform to the octet rule i.e. in the case of carbon, 8 electrons in its outermost shell. An easy way to remember the number of valence electrons of an atom is by following the periodic table where each group represents a particular valency number.
Group 1 = +1 Group 2 = +2 Group 13 = +3 Group 14 = +4/-4
Group 15 = -3 Group 16 = -3 Group 17 = -1 Group 18 = 0
A few rules while making the Lewis Dot Structure for a compound involves first, drawing the dot structure for an atom, second, connecting the atoms with more than one bond, third, connecting the atoms with one bond, and finally checking if each atom follows the octet rule.
Examples of Lewis Structures –