Lewis Acid Definition:
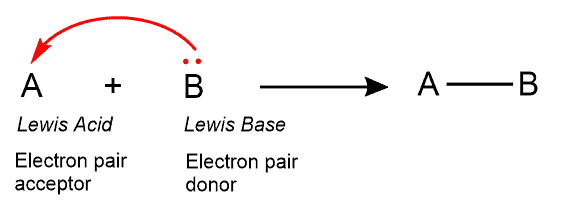
A Lewis acid is a compound that acts as an electron-pair acceptor.
Lewis Acid Explained:
The Lewis theory contrary to the Bronsted-Lory concept uses electrons instead of proton transfers to identify acids and bases. To put it into a simplified way, a Lewis acid takes accepts a pair of electrons while a Lewis base donates a pair of electrons bases gives them away.
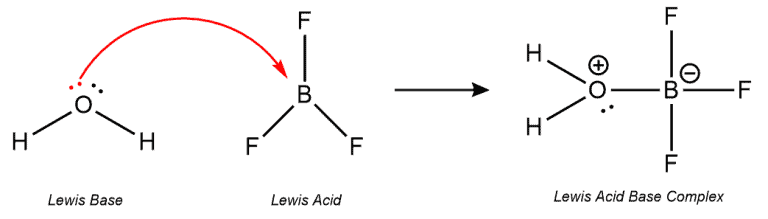
Examples –