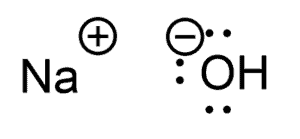Ionic Bond Definition:
An ionic bond is a bond that is formed due to the force of attraction between two oppositely charged ions. This is observed when the difference in electronegativity between the bonding atoms is greater than 1.7.
Ionic Bond Explained:
The ionic bond is one of the two basic types of chemical bonds in which atoms transfer electrons to each other. The bonding requires at least one electron donor and one electron acceptor atom i.e. atoms with a high difference in electronegativity. This leads to the formation of two oppositely charged ions. The force of attraction between the ions causes the formation of a bond. Notice that is different from covalent/polar covalent bonds in which the electrons are shared.
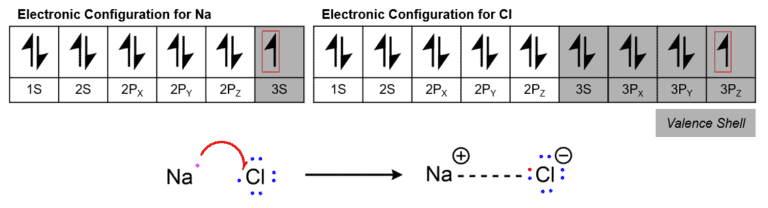
While studying ionic bonds, we will find that the metal atom usually acts as the electron donor and a non-metal atom usually acts as the electron acceptor. Judging by the Octet Rule, the metal atoms have few electrons in the valence shell and can readily donate an electron to achieve the nearest noble gas electronic configuration. The same goes for the non-metal atom, except the fact that it readily accepts the electron to achieve the nearest noble gas electronic configuration. Given the importance of achieving the Octet, it is possible that the atom gains or loses more than one electron the exact number of which is represented as the charge on the atom. Positive if it gives up the electron and negative if it accepts the electron. The net charge on the compound must always be zero.
Examples – NaCl. Here, the Na molecule donates an electron from its valence shell in order to achieve octet configuration while the chlorine atom accepts one electron into its valence shell to achieve the same.
Other Examples – NaOH