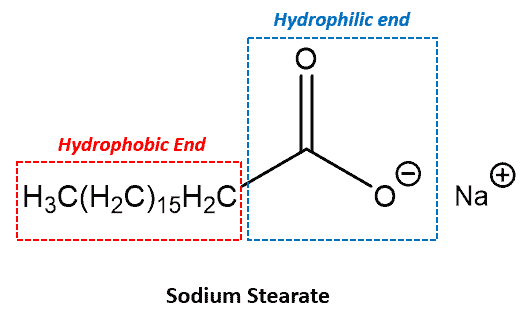
Hydrophobic Definition:
Hydrophobic is the name given to a non-polar group that does not interact favorably with water.
Hydrophobic Explained:
The concept originates from the observation that “like dissolves like”. That is, a polar molecule is soluble dissolves in a polar solvent while a non-polar molecule is soluble in a non-polar solvent. Hence, non-polar molecules that are repelled by water is a polar molecule) are called hydrophobic.
Examples – Oil, Wax.