Heterolytic Bond Cleavage Definition:
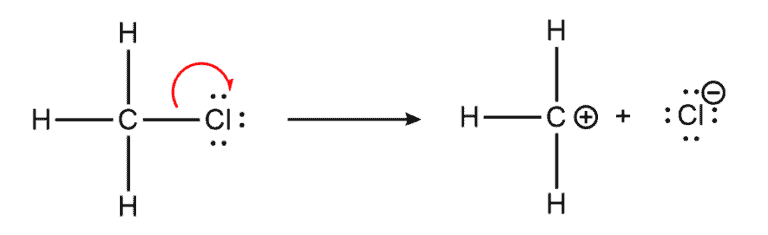
From Bond cleavage. In a heterolytic bond cleavage, the bonding pair of electrons is split unevenly between the products. The cleaving generates two charged product species or ions.
Heterolytic Bond Cleavage Explained:
Generalization

Notice: The bond is cleaved such that one of the bonding species takes both original bonding electrons from the other species.
Example – Heterolysis of a carbon-leaving group bond that gives a carbocation and a negatively charged ion as products. This is also the rate-limiting step in the SN1 and E1 mechanisms.