Degenerate Orbitals Definition:
Degenerate orbitals are orbitals that have the same energy.
Degenerate Orbitals Explained:
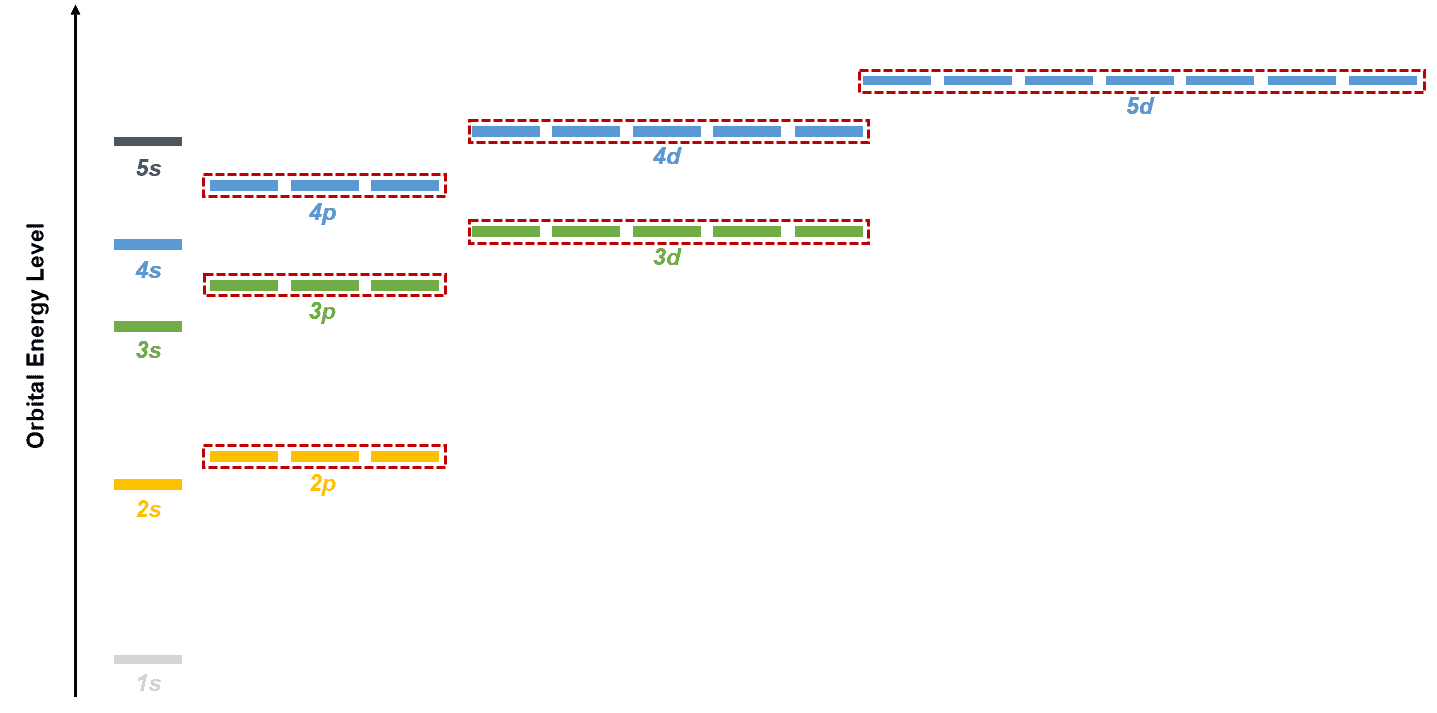
After we understanding atomic orbitals, we must also understand the energy states of these orbitals. A basic visualization of these energy states is as shown below. Notice that few sets of orbitals are circled in red. These orbitals have the same energy is called degenerate.
For example, for the 2p shell, the 2px, 2py, and 2pz orbitals are degenerate. We shall revisit this concept when discussing principles like the Aufbau principle, Hund’s rule, and Pauli’s exclusion principle that discuss the electron configuration of atoms.