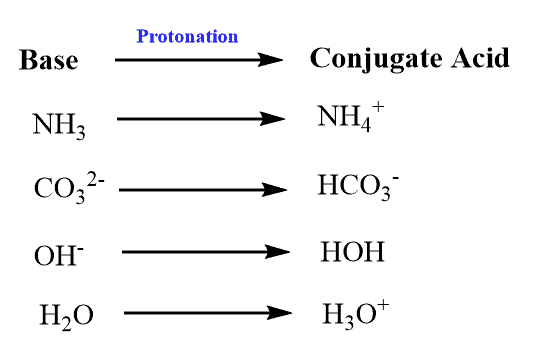
Conjugate Acid Definition:
A conjugate acid, in an acid-base reaction, is the product that results when a base is protonated.
Conjugate Acid Explained:
A conjugate acid is, therefore, a base with an extra hydrogen atom added to it.
- A strong conjugate acid results from a weak base.
- A weak conjugate acid results from a strong base.
Examples –