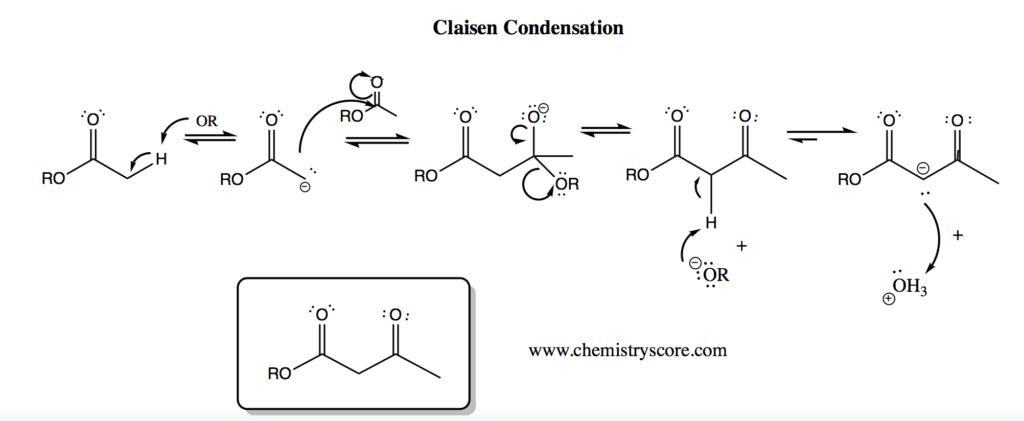
Claisen Condensation Definition:
Claisen condensation is defined as a nucleophilic acyl substitution reaction in which the nucleophile is an ester enolate and the electrophile is an ester.
Claisen Condensation Explained:
The carbon bonds form between two esters or one ester with another carbonyl compound while there is a strong base which when combined creates a reaction. The mechanism by which this reaction is carried out consists of four stages:
- Deprotonation of the alpha position for an ester enolate.
- The enolate is now a good nucleophile and attacks an ester.
- The intermediate stabilizes by reforming a carbonyl group, i.e. removing an alkoxide ion.
- The alpha position is again deprotonated to form a double, stable enolate.