Brønsted-Lowry Base Definition:
A Bronsted-Lowry base is a compound that serves as a proton(H+) acceptor.
Brønsted-Lowry Base Explained:
In a Bronsted-Lowry Base, the compound is able to accept a proton
When we talk about the Bronsted-Lowry reactions, it’s important to think conjugates.
- A strong base will have a weak conjugate acid.
- A weak base will have a strong conjugate acid
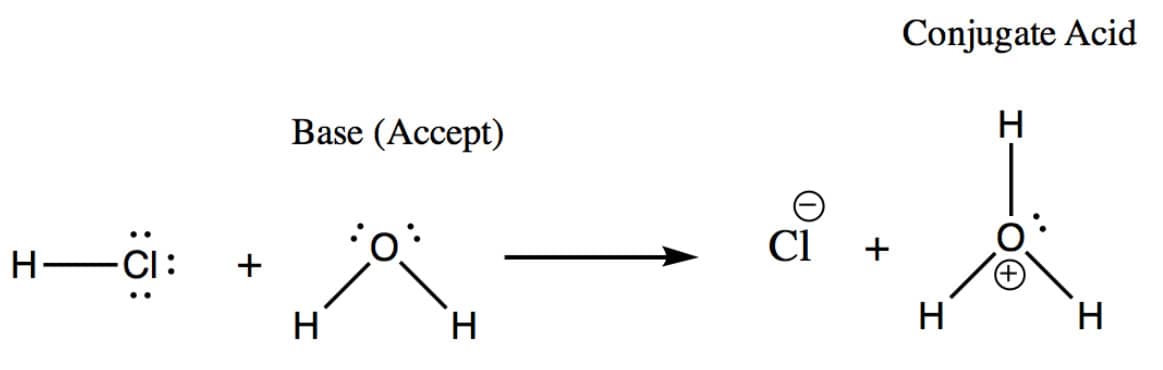
There are several lists that denote which acids are strong and which are weak. It’s important to know these, especially when more complex reactions are taking place.