Bimolecular Definition:
A bimolecular reaction, in chemistry, refers to any reaction step occurring as a result of two reactants. For example, reactant [A] and reactant [B] undergoing a chemical reaction to yield product [C].
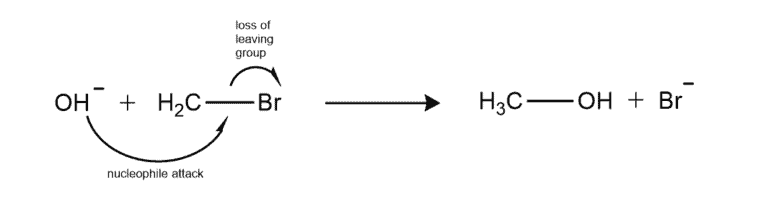
However, in terms of organic chemistry, a bimolecular reaction refers to a bimolecular nucleophilic substitution, also known as the SN2 mechanism.
Bimolecular Explained:
The SN2 Reaction (S-Substrate, N- Nucleophile, 2 – to denote bimolecular) is a characteristic reaction mechanism in which the rate of reaction is linearly dependent on the concentration of two different compounds. i.e the rate is directly proportional to the rate constant times the conc. of the substrate times the conc. of the nucleophile, both raised to the power of 1. In this mechanism, a nucleophile attaches to the central carbon of an organic halide (the substrate), causing the departure of the leaving group. Hence, we call this step a bimolecular step since it involves two chemical entities.
For example –